|
Return
to
Index Page
Activity:
Herbicide
(sulfonylurea)
Structure:
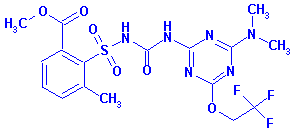
Adverse Effects:
Anemia
Blood
Body Weight Decrease
Bone
Brain
Cancer: Possible Human Carcinogen - BLOOD,
TESTES, LIVER
Endocrine: Testicular
Gastrointestinal
Genotoxicity
Liver
Sciatic nerve
Teratogenic
Environmental
Triflusulfuron
is a sulfonylurea herbicide. It will be marketed as a 50%
dry flowable product under the name UPBEET ¨ 50 DF. Triflusulfuron-methyl
was first registered in 1993 in Belgium and France, and
subsequently in Germany, the Netherlands, Finland and Italy.
In Europe, the end-use product is known as SAFARI and DEBUT.
In the U.S., the product was registered in 1996...
Ref:
Dec 3, 1999 - Report on Triflusulfuron methyl. Regulatory
Note REG99-03. Pest Management Regulatory Agency, Health
Canada, Ottawa.
http://www.fluorideaction.org/pesticides/triflusulfuron.methy.canada.pdf
As
of February 17, 2005, residues of Triflusulfuron-methyl
are permitted in or on 3 food commodities.
See list at:
http://www.fluorideaction.org/pesticides/mrl.triflusulfuron-methyl.htm
|
Anemia
(click on for all fluorinated
pesticides)
Two 90-day studies
were conducted in the rat. In one study, rats were fed dosages
of 6.2, 127, 646, or 965 mg/kg/day (males) or 7.54, 150, 774,
or 1,070 mg/kg/day (females). Triflusulfuron methyl exhibited
subchronic toxicity at dietary concentrations of 2,000 ppm (127
and 150 mg/kg/day for males and females) or greater in the form
of decreased body weights, decreased body weight gains, decreased
food efficiency, increased mean relative liver weights, and regenerative
anemia. The NOAEL was 6.2 mg/kg/day
(males) and 7.54 mg/kg/day (females).
Ref: Federal Register: August 8, 2001 (Volume
66, Number 153)] [Notices] [Page 41593-41597]. Notice of Filing
a Pesticide Petition to Establish a Tolerance for a Certain Pesticide
Chemical in or on Food.
http://www.fluoridealert.org/pesticides/Triflusulfuron.M.FR.Aug8.01.htm
Blood
(click on for all fluorinated
pesticides)
-- Carcinogenicity
rats NOAEL = 2.44 mg/kg/day LOAEL
= 30.6 mg/kg/day based on decreased body
weight and body weight gain, alteration
in hematology (mainly males) and increased
incidences of interstitial cell hyperplasia in the testes. (Possible)
evidence of carcinogenicity
Ref: Federal Register: June 12, 2002 (Volume
67, Number 113). Triflusulfuron Methyl; Pesticide Tolerance. Final
Rule.
http://www.fluorideaction.org/pesticides/triflusulfuron.m.fr.june.02.htm
CHRONIC STUDIES: Triflusulfuron methyl Chronic Feeding Study
in Rats - Non-oncogenic in female rats. Increased incidence of
Leydig cell adenomas were observed in males following chronic
and excessive exposures at 750 and 1500 ppm. The
NOELs for male and female rats were
100 and 750 ppm, respectively.
These were based on increased adenomas and
reduced circulating red cell mass among higher-dosed males and
reduced body weights and increased incidence or severity of species-
and age-specific effects among male and female rats at higher
doses...
Ref: Nov 2001 - Material Safety Data Sheet
for DuPont "UPBEET" HERBICIDE.
http://www.fluorideaction.org/pesticides/triflusulfuron.methyl.msds.pdf
In another 90-day subchronic
study, dogs were fed dosages of 3.87, 146.1, or 267.6 mg/kg/day
(males) or 3.72, 159.9, or 250.7 mg/kg/day (females). Triflusulfuron
methyl was found to be hepatotoxic at 4,000 ppm (146.1 mg/kg/day
males and 159.9 mg/kg/day females), and greater elevated hepatic
enzyme levels and postmortem evidence, including elevation in
liver weights and microscopic evidence of bile stasis. Other microscopic
findings considered to be treatment related were testicular atrophy
and decreased testicular weights and hypercellularity
of the sternal and femoral bone marrow, with a corresponding increase
in reticulocyte and leukocyte counts seen in the high-dose
males and females. Based on the microscopic findings in the liver
and testes of the 4,000 ppm and greater treated animals, the NOAEL
was 3.87 mg/kg/day (males) and 3.72 mg/kg/day (females).
Ref: Federal Register. August 8, 2001. [PF-1036;
FRL-6795-4]
http://www.fluoridealert.org/pesticides/Triflusulfuron.M.FR.Aug8.01.htm
Body
Weight Decrease
(click
on for all fluorinated pesticides)
-- TERATOLOGY, RAT. 51974-030; 119842;
"Teratogenic Study of DPX-66037-24
in Rats" (Author: C.A. Mebus; E.I. du Pont de Nemours
& Company, Inc., Newark, DE, Project No. HLR 525-91, 11/1/91);
DPX-66037-24 (95.6% Triflusulfuron-methyl); 0, 30, 120, 350, 1000
mg/kg/day oral gavage; 25 Crl: CD BR female rats/dose; observations-
maternal effects; five animals died due to dosing injuries; significant
decreases in maternal weight changes and feed consumption were
observed; fetal effects; significantly increased average
number of malformed fetuses were observed, the majority of fetal
malformations occurred in one fetus, when individual end points
of developmental evaluation criteria were grouped a significant
increase was noted in the 350 and 1000 mg/kg dose groups primarily
due to retarded renal development and partial ossification of
skulls, sternebra or vertebra; no adverse effect; Maternal NOEL
= 120 mg/kg (based on decreased maternal weight gain and feed
consumption), Developmental NOEL = 120number of malformed fetuses,
retarded fetal development)
-- TERATOLOGY, RABBIT. 51974-031;
119843; "Teratogenic Study of DPX-66037-24
in Rabbits" (Author: S.M. Murray; E.I. du Pont de
Nemours & Company, Inc., Newark, DE, Project No. HLR 575-91,
10/28/91); DPX-66037-24 (95.6% Triflusulfuron-methyl);
0, 15, 90, 270, 800 mg/kg/day oral gavage; 20 Hra:(NZW)SPF female
rabbits/dose; observations- maternally toxic at doses of 90 mg/kg
and greater, significant increased dose-related
mortalities and spontaneous abortions occurred at levels
of 270 and 800 mg/kg, during the dosing period the highest two
dose groups showed clinical signs of reduced fecal output, reduced
fecal size or diarrhea; significant decreases
in maternal weight changes and feed consumption were also
observed; histopathological changes were noted primarily in the
800 mg/kg group and included gastric mucosal ulcerations and digestive
tract gaseous distension; death losses at
800 mg/kg were extreme and data from remaining dams were markedly
different therefore these data were excluded from statistical
analysis; no physiological or developmental fetal effects were
observed at any dose level; no adverse effect; Maternal NOEL =
15 mg/kg (based on animal death, abortions,
decreased maternal weight gain and food consumption), =
800 mg/kg (based on no adverse affects).
-- 51974-058; 127247; "Combined Chronic
Toxicity/Oncogenicity Study with DPX-66037-24 Two-Year
Feeding Study in Rats" (Author: L.B. Biegel; E.I.
du Pont de Nemours & Company, Inc., Newark, DE; Project No.
HLR 3-93; 5/6/93); DPX-66037-24 (95.6% triflusulfuron-methyl);
0, 10, 100, 750, 1,500 ppm (averaged intake (M) 0, 0.406, 4.49,
30.6, 64.5 and (F) 0, 0.546, 5.47, 41.5, 87.7 mg/kg/day, respectively)in
diets; 62 rats/sex/dose; observations- statistically
significant decreased body weights and body weight gains of rats
in the 1,500 ppm group; in females fed 750 and 1500 ppm
a decreased incidence of mammary masses was noted when compared
to controls, in the highest dosed females statistically significant
increases of sciatic nerve myelin/axon degeneration occurred,
the 1500 ppm males had no increased incidence of this lesion but
showed increased lesion severity; males in 750 and 1500 ppm groups
had decreased erythrocyte counts and mean serum triglyceride concentrations;
high dosed males showed decreased serum calcium concentrations,
an increase in the absolute and relative testes weight and statistically
significant increases of both Leydig cell hyperplasia and adenomas;
NOEL(M)= 100 ppm (based on decreased erythrocyte counts, decreased
body weights, decreased body weight gain, increased severity of
sciatic nerve lesions, Leydig cell hyperplasia and Leydig cell
adenomas); NOEL(F)= 750 ppm (based on decreased body weight and
increased incidence of sciatic nerve degeneration); Possible Adverse
Effect: Leydig cell hyperplasia and adenomas; Acceptable. (Miller,
12/13/93)
-- REPRODUCTION, RAT ** 51974-057;
126843; "Reproductive and Fertility
Effects with DPX-66037-24 Multigeneration Reproduction Study in
Rats" (Author: M. E. Hurtt; E.I. du Pont de Nemours
& Company, Inc., Newark, DE, Project No. HLR 231-92, 4/20/93);
DPX-66037-24 (95.6% Triflusulfuron-methyl); 0, 10, 100, 750, 1,500
ppm dietary; 30 Crl: CD BR rats/dose both P1 and F1; observations-
no dose related mortalities were observed, statistically significant
lower body weights and body weight gains were observed in P1 (two
highest doses) and F1 (1500 ppm) males, body weight effects were
accompanied by statistically significant decreases in food consumption
(1500 ppm group), in female P1 rats statistically
significant lower mean body weight gains were noted (750,
1500 ppm groups) during premating, statistically
significant lower mean body weights during gestation and lactation
(two highest groups) were also observed, statistically significant
decreases in overall food consumption was present in F1 female
rats during premating (750, 1500 ppm groups), no additional
statistically significant differences were found in other adult
parameters; compound related decreases in mean F1 and F2 pup weights
(highest two dose groups) were observed but were not statistically
significant, there were no significant differences in any other
clinical observations in the F1 and F2 pups; generally gross necropsy
and histomorphologic findings of adults and offspring were few
and were not considered treatment related; Adult and Developmental
NOEL = 100 ppm of a.i. (based on decreased body weight, body weight
gain, food efficiency and decreased pup weights); Acceptable (Miller,
11/18/93).
-- Subchronic Neurotoxicity, Rat **51974-099;
147828; "Subchronic Neurotoxicity Study
of DPX-66037-24 (Triflusulfuron methyl) Administered Orally
via the Diet to Crl:CDBR VAF/Plus Rats" J.A. Foss; E.I. du
Pont de Nemours & Company, Newark, DE; Project No. DuPont
HLO 127-93; 9/29/94; DPX-66037-24 (95.6%, Triflusulfuron-methyl);
male doses (consumed mean doses [mg/kg/day] range) 0, 100 (4.4-9.7),
750 (72.1-32.8), 1500 (143.1-66.5), 3000 (272.1-133.8) ppm in
diets, female doses (consumed mean doses [mg/kg/day] range) 0,
100 (5.5-9.7), 750 (67.7-40.0), 1500 (136.2-79.0), 3000 (258.3-163.4)
ppm in diets ; 11 rats/sex/dose; observations-No specific evidence
of behavioral or histological neurotoxicity at any level. The
750 ppm and higher dietary concentrations
significantly reduced body weight gains and feed consumption values
and the two highest concentrations significantly reduced the average
body weights in females. The 3000 ppm concentration significantly
reduced body weight gains and showed a trend to reduce average
body weights and feed consumption values in male rats. NOEL(F)=
100 ppm (based on decreased body weight gain); NOEL(M)= 1500 ppm
(based on decreased body weight gain); No adverse effects.
-- Rabbit 21-Day Repeated Dosing Dermal
Toxicity Study 51974-093; 147897; Subacute Dermal Toxicity;
822; Rabbit; "Repeated Dose Dermal Toxicity: 21-Day Study
with DPX-66037-24 (Triflusulfuron Methyl) in Rabbits"; S.A.
MacKenzie; E.I. du Pont de Nemours and Co., Haskell Lab. for Tox.
and Ind. Med., Newark, DE; Laboratory Report No. HRL 552-93; 9/27/93
; Triflusulfuron Methyl (DPX-66037-24, 95.6% purity); 0, 50, 300,
1000 mg/kg applied dermally 6 hrs/day for 21 days ; 822; 5 New
Zealand White rabbits/sex/dose; observations- There were no test
article related mortalities or clinical signs of systemic toxicity.
Slight or mild erythema was observed in animals of all groups
(including controls). Mild skin trauma was noted in a few animals
but was not considered compound-related. Microscopic skin lesions
observed in both treated and control groups included, minimal
to mild inflamation and acanthosis. Lesions were attributed to
testing procedure. Statistically significant
differences in mean body weight gain were observed over a few
intervals in female rabbits but did not exibit a dose-response
and were not considered compound related. No other compound
related changes were noted any other parameter including body
weight or body weight gain, food consumption, hematology or clinical
chemistry analysis. Systemic and Dermal NOEL(M/F)= NOAEL(M/F)=1000
mg/kg b.w. (based on no effects at HDT).
-- Dog Subchronic Dietary Toxicity Study
51974-029; 119838; "A Subchronic (3-month)
Toxicity Study of DPX-66037-24 in the Dog Via Dietary Administration"
(Author: J.E. Atkinson; Bio/dynamics, Inc., East Millstone, NJ,
Project No. 91-3653, 12/20/91); DPX-66037-24(95.6% triflusulfuron-methyl);
0, 100, 4,000 and 8,000 ppm (averaged intake (M) 3.9, 146.1, 267.6
and (F) 3.7, 159.9 and 250.7 mg/kg/day) in diets; 4 Beagle dogs/sex/dose;
observations- two high dose females were
sacrificed in extremis showing marked body weight loss with
low food consumption and frank anemia, other high dose animals
exhibited little to no weight gain with food consumption slightly
decreased in the early weeks of the study; decreased erythrocyte
count and associated hematocrit and hemoglobin levels were noted
at 1 1/2 and 3 months with a compensatory elevated reticulocyte
count; hepatotoxcity was demonstrated by elevated levels of serum
aspartate aminotransferase, serum alanine aminotransferase, alkaline
phosphatase and elevated liver weights and microscopic evidence
of bile stasis in the 4,000 and 8,000 ppm groups;
in mid and high dose males testicular atrophy and decreased testicular
weights were characterized by aspermatogenesis, decreased seminiferous
tubule thickness and germinal epithelial cytoplasmic vacuolation;
sternal and femoral bone marrow hypercellularity was seen in high
dose animals; NOEL(M/F)= 100 ppm (based on microscopic findings
in the liver and testes); Possible Adverse Effect: testicular
atrophy, aspermatogenesis.
Reference: Nov 18, 2005 - Summary of Toxicology Data. California
Environmntal Protection Agency. Department of Pesticide Regulation.
Medical Toxicology Branch.
http://www.fluoridealert.org/pesticides/triflusulfuron-m.ca.epa.2005.pdf
In a rat reproduction study (2-generation, 1 litter/generation),
the NOEL for systemic (parental) toxicity was 5.81 mg/kg bw/d
based on lower body weights and food consumption. The NOEL for
reproductive toxicity was 44 mg/kg bw/d (750 ppm) based on lower
pup body weights and histopathological effects
on the cerebellum consistent with undernutrition
(decreased cellularity in the internal granular
layer and increased cellularity in the external germinal layer)
in the 2500 ppm group. In the rat developmental toxicity
study, the NOEL for maternal toxicity was 120 mg/kg bw/d (based
on reduced body-weight gain during
dosing period at 350 and 1000 mg/kg bw/d) and for developmental
toxicity was 120 mg/kg bw/d (based on delayed
ossification). There was no evidence of teratogenicity.
In the rabbit developmental toxicity study, the NOEL for maternal
toxicity was 15 mg/kg bw/d based on decreased
body-weight gain during the treatment period at 90 mg/kg
bw/d and clinical signs of toxicity at 270 mg/kg bw/d. The NOEL
for developmental toxicity was 90 mg/kg bw/d, based on abortions
at 270 mg/kg bw/d. There was no evidence of any teratogenic potential
of triflusulfuron-methyl in the rabbit up to 800 mg/kg bw/d.
Ref: Dec 3, 1999 - Report on Triflusulfuron
methyl. Regulatory Note REG99-03. Pest Management Regulatory Agency,
Health Canada, Ottawa.
http://www.fluorideaction.org/pesticides/triflusulfuron.methy.canada.pdf
-- Reproduction and fertility effects. Parental/Systemic NOAEL
= 5.81/7.75 (m/f) mg/kg/day LOAEL = 44/58 mg/kg/day based on decreased
body weight, decreased body weight gain, decreased food
consumption, and decreased- food efficiency. Reproductive NOAEL
= 89.5/115 (m/f) mg/kg/ day based on the absence of reproductive
effects at the highest dose tested (HDT). LOAEL = 3115 mg/kg/day.
Offspring NOAEL = 5.81/7.75 (m/f) mg/kg/day LOAEL = 44/58 (m/f)
mg/kg/day based on decreased F1 pup body
weight on days 14 and 21 due to exposure via milk and in
the diet.
-- Subchronic neurotoxicity screening battery. NOAEL = 92.7/7.1
(m/f) mg/kg/day LOAEL = 186.2/51.6 (m/f) mg/kg/day based
on decreased body weight and body weight gain.
Ref: Federal Register: June 12, 2002 (Volume
67, Number 113). Triflusulfuron Methyl; Pesticide Tolerance. Final
Rule.
http://www.fluorideaction.org/pesticides/triflusulfuron.m.fr.june.02.htm
Two 90-day studies
were conducted in the rat. In one study, rats were fed dosages
of 6.2, 127, 646, or 965 mg/kg/day (males) or 7.54, 150, 774,
or 1,070 mg/kg/day (females). Triflusulfuron methyl exhibited
subchronic toxicity at dietary concentrations of 2,000 ppm (127
and 150 mg/kg/day for males and females) or greater in the form
of decreased body weights, decreased
body weight gains, decreased food efficiency, increased
mean relative liver weights, and regenerative anemia.
The NOAEL was 6.2 mg/kg/day (males) and 7.54 mg/kg/day (females).
Ref: Federal Register: August 8, 2001 (Volume
66, Number 153)] [Notices] [Page 41593-41597]. Notice of Filing
a Pesticide Petition to Establish a Tolerance for a Certain Pesticide
Chemical in or on Food.
http://www.fluoridealert.org/pesticides/Triflusulfuron.M.FR.Aug8.01.htm
-- Chronic toxicity
rodents NOAEL = 2.44 mg/kg/day LOAEL
= 30.6 mg/kg/day based on decreased body
weight and body weight gain, alteration
in hematology (mainly males) and
increased incidences of interstitial cell hyperplasia in testes.
-- Carcinogenicity rats NOAEL = 2.44
mg/kg/day LOAEL = 30.6 mg/kg/day based on decreased
body weight and body weight gain, alteration
in hematology (mainly males) and increased incidences of interstitial
cell hyperplasia in the testes. (Possible) evidence of carcinogenicity
Ref: Federal Register: June 12, 2002 (Volume
67, Number 113). Triflusulfuron Methyl; Pesticide Tolerance. Final
Rule.
http://www.fluorideaction.org/pesticides/triflusulfuron.m.fr.june.02.htm
Bone
(click on for all fluorinated
pesticides)
In a rat reproduction study (2-generation,
1 litter/generation), the NOEL for systemic (parental) toxicity
was 5.81 mg/kg bw/d based on lower body weights and food consumption.
The NOEL for reproductive toxicity was 44 mg/kg bw/d (750 ppm)
based on lower pup body weights and histopathological
effects on the cerebellum consistent with undernutrition (decreased
cellularity in the internal granular layer and increased cellularity
in the external germinal layer) in the 2500 ppm group. In the
rat developmental toxicity study, the NOEL for maternal toxicity
was 120 mg/kg bw/d (based on reduced body-weight gain during dosing
period at 350 and 1000 mg/kg bw/d) and for developmental
toxicity was 120 mg/kg bw/d (based on delayed
ossification). There was no evidence of teratogenicity...
Ref: Dec 3, 1999 - Report on Triflusulfuron
methyl. Regulatory Note REG99-03. Pest Management Regulatory Agency,
Health Canada, Ottawa.
http://www.fluorideaction.org/pesticides/triflusulfuron.methy.canada.pdf
In another 90-day subchronic
study, dogs were fed dosages of 3.87,
146.1, or 267.6 mg/kg/day (males) or 3.72, 159.9, or 250.7 mg/kg/day
(females). Triflusulfuron methyl was found to be hepatotoxic at
4,000 ppm (146.1 mg/kg/day males and 159.9 mg/kg/day females),
and greater elevated hepatic enzyme levels and postmortem evidence,
including elevation in liver weights and microscopic evidence
of bile stasis. Other microscopic findings considered to be treatment
related were testicular atrophy and decreased testicular weights
and hypercellularity of the sternal and
femoral bone marrow, with a corresponding increase in reticulocyte
and leukocyte counts seen in the high-dose males and females.
Based on the microscopic findings in the liver and testes of the
4,000 ppm and greater treated animals, the NOAEL was 3.87 mg/kg/day
(males) and 3.72 mg/kg/day (females).
Ref: Federal Register. August 8, 2001. [PF-1036;
FRL-6795-4]
http://www.fluoridealert.org/pesticides/Triflusulfuron.M.FR.Aug8.01.htm
51974-030; 119842; "Teratogenic Study of DPX-66037-24 in
Rats" (Author: C.A. Mebus; E.I. du Pont de Nemours &
Company, Inc., Newark, DE, Project No. HLR 525-91, 11/1/91); DPX-66037-24
(95.6% Triflusulfuron-methyl); 0, 30, 120, 350, 1000 mg/kg/day
oral gavage; 25 Crl: CD BR female rats/dose;
observations- maternal effects; five animals died due to dosing
injuries; significant decreases in maternal weight changes and
feed consumption were observed; fetal effects; significantly increased
average number of malformed fetuses were observed, the majority
of fetal malformations occurred in one fetus, when individual
end points of developmental evaluation criteria were grouped a
significant increase was noted in the 350 and 1000 mg/kg dose
groups primarily due to retarded renal development and
partial ossification of skulls, sternebra or vertebra;
no adverse effect; Maternal NOEL = 120 mg/kg (based on decreased
maternal weight gain and feed consumption), Developmental NOEL
= 120 number of malformed fetuses, retarded
fetal development).
Reference: Nov 18, 2005 - Summary of Toxicology
Data. California Environmntal Protection Agency. Department of
Pesticide Regulation. Medical Toxicology Branch.
http://www.fluoridealert.org/pesticides/triflusulfuron-m.ca.epa.2005.pdf
Brain
(click
on for all fluorinated pesticides)
In a rat reproduction study (2-generation, 1 litter/generation),
the NOEL for systemic (parental) toxicity was 5.81 mg/kg bw/d
based on lower body weights and food consumption. The NOEL for
reproductive toxicity was 44 mg/kg bw/d (750 ppm) based on lower
pup body weights and histopathological effects
on the cerebellum consistent with undernutrition (decreased cellularity
in the internal granular layer and increased cellularity in the
external germinal layer) in
the 2500 ppm group...
Ref: Dec 3, 1999 - Report on Triflusulfuron
methyl. Regulatory Note REG99-03. Pest Management Regulatory Agency,
Health Canada, Ottawa.
http://www.fluorideaction.org/pesticides/triflusulfuron.methy.canada.pdf
Cancer:
Possible Human Carcinogen - BLOOD, TESTES, LIVER
(click
on for all fluorinated pesticides)
Group C--Possible Human Carcinogen.
Reviewed 5/ 28/ 96.
Ref: List of Chemicals Evaluated for Carcinogenic
Potential. Science Information Management Branch, Health Effects
Division, Office of Pesticide Programs, U. S. Environmental Protection
Agency. March 15, 2002.
http://www.biomuncie.org/chemicals_evaluated_for_carcinog.htm
-- Carcinogenicity
rats NOAEL = 2.44 mg/kg/day LOAEL = 30.6 mg/kg/day based on decreased
body weight and body weight gain, alteration in hematology
(mainly males) and increased incidences of
interstitial cell hyperplasia in the testes. (Possible)
evidence of carcinogenicity
-- Carcino-genicity mice NOAEL = 14.6 mg/kg/day LOAEL = 349 mg/kg/day
based on increased liver weight and increased hepatic
cell tumors (adenomas and/or carcinomas combined.
(Possible) evidence of carcinogenicity
Ref: Federal Register. June 12, 2002. Triflusulfuron
Methyl; Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Triflusulfuron.M.FR.June.02.htm
Endocrine:
Testicular
(click on for all fluorinated pesticides)
-- 90-Day oral toxicity
in nonrodents. NOAEL = 3.9/3.7 (m/f)
mg/kg/day LOAEL = 146.9/159.9 (m/f) mg/kg/day based on decreased
mean body weight and body weight gain, decreased hematocrit, hemoglobin,
RBC`s, SGOT, SGPT, ALP, absolute and relative liver and testes
weight; microscopic abnormalities of the liver and testes.
-- Chronic toxicity rodents NOAEL
= 2.44 mg/kg/day LOAEL = 30.6 mg/kg/day based on decreased
body weight and body weight gain, alteration
in hematology (mainly males) and
increased incidences of interstitial cell hyperplasia in testes.
-- Carcinogenicity rats NOAEL = 2.44
mg/kg/day LOAEL = 30.6 mg/kg/day based on decreased
body weight and body weight gain, alteration
in hematology (mainly males) and
increased incidences of interstitial cell hyperplasia in the testes.
(Possible) evidence of carcinogenicity
-- Special studies: In vivo and in vitro mechanic studies. The
purpose of these studies was to investigate the mechanism of
Leydig cell tumor induction in the testes of male rats. A dose-dependent
decrease in aromatase enzyme activity was seen in vitro, but was
inconclusive in vivo.
Ref: Federal Register: June 12, 2002 (Volume
67, Number 113). Triflusulfuron Methyl; Pesticide Tolerance. Final
Rule.
http://www.fluorideaction.org/pesticides/triflusulfuron.m.fr.june.02.htm
Dog Subchronic Dietary Toxicity Study
51974-029; 119838; "A
Subchronic (3-month) Toxicity Study of DPX-66037-24 in the Dog
Via Dietary Administration"
(Author: J.E. Atkinson; Bio/dynamics, Inc., East Millstone,
NJ, Project No. 91-3653, 12/20/91); DPX-66037-24(95.6% triflusulfuron-methyl);
0, 100, 4,000 and 8,000 ppm (averaged intake (M) 3.9, 146.1, 267.6
and (F) 3.7, 159.9 and 250.7 mg/kg/day) in diets; 4 Beagle dogs/sex/dose;
observations- two high dose females were sacrificed in extremis
showing marked body weight loss with low food consumption and
frank anemia, other high dose animals exhibited little to no weight
gain with food consumption slightly decreased in the early weeks
of the study; decreased erythrocyte count and associated hematocrit
and hemoglobin levels were noted at 1 1/2 and 3 months with a
compensatory elevated reticulocyte count; hepatotoxcity was demonstrated
by elevated levels of serum aspartate aminotransferase, serum
alanine aminotransferase, alkaline phosphatase and elevated liver
weights and microscopic evidence of bile stasis in the 4,000 and
8,000 ppm groups; in mid and high dose males
testicular atrophy and decreased testicular weights were characterized
by aspermatogenesis, decreased seminiferous tubule thickness and
germinal epithelial cytoplasmic vacuolation; sternal and
femoral bone marrow hypercellularity was seen in high dose animals;
NOEL(M/F)= 100 ppm (based on microscopic findings in the liver
and testes); Possible Adverse Effect:
testicular atrophy, aspermatogenesis.
Reference: Nov 18, 2005 - Summary of Toxicology
Data. California Environmntal Protection Agency. Department of
Pesticide Regulation. Medical Toxicology Branch.
http://www.fluoridealert.org/pesticides/triflusulfuron-m.ca.epa.2005.pdf
-- Groups of 62 rats/sex were fed
diets containing 0, 10, 100, 750 or 1500 ppm DPX-66037- 24 (95.6%
purity) (equal to 0.406, 4.06, 30.6 or 64.5 mg/kg bw/d for males
and 0.546, 5.47, 41.5 and 87.7 mg/kg bw/d for females) for 22
months. The NOEL for this study was 100 ppm (4.06 mg/kg bw/d).
At 750 ppm, body weights and body-weight
gains were lower than controls in both sexes and males had lower
erythrocyte counts than controls at most time points and an increased
incidence of Leydig cell hyperplasia compared to controls. The
incidence of myelin/axonal degeneration of the sciatic nerve was
increased compared to controls in the 1500-ppm group of females
(25/48 versus [vs] 42/49). Triflusulfuron-methyl was
oncogenic in male rats under the conditions of this study, with
an increased incidence of Leydig cell adenomas in the 750- and
1500-ppm groups compared to controls.
-- Possible mechanisms of Leydig
cell tumour development induced by triflusulfuron-methyl
were studied. Ten Crl:CD ¨ BR male rats/group
were treated by gavage for 15 days with 0, 1000, 1500 or 2000
mg/kg bw DPX-66037-24 (95.6% purity) in corn oil. An additional
group of 10 control rats were pair-fed to the 2000 mg/kg bw group.
Satellite groups of 10 rats treated with 0 or 2000 mg/kg bw DPX-66037
were given human chorionic gonadotropin (hCG) one hour before
sacrifice. All treated groups had lower body weights and food
consumption than controls. Absolute and relative weights of the
prostate, seminal vesicles and coagulating glands were lower than
controls. Serum estradiol was statistically significantly lower
than controls. There were slight, but not statistically
significant, increases in luteinizing hormone (LH), follicule-stimulating
hormone (FSH) and prolactin in the treated groups compared to
controls. In the rats treated with hCG, the 2000 mg/kg bw/d group
had increased testosterone and lower estradiol than the controls.
-- Isolated Leydig cells from 11-week old male
rats were exposed to 0, 0.1, 0.5, 1.0, 10, 100 or 1000
FM DPX-66037-24 for two hours. Three cultures from each dose were
then exposed to two international units of hCG. No effect on hormone
levels was observed in the hCG-treated cultures. In
the triflusulfuron-methyl-only cultures, testosterone was significantly
increased (198%) compared to controls, and
estradiol was decreased. Triflusulfuron-methyl
appears to inhibit the conversion of testosterone to estradiol
by aromatase in vitro; the in vivo results are inconclusive.
-- In long-term rodent dietary studies,
the NOEL for chronic (18-mo) systemic toxicity in mice
was 20.9 mg/kg bw/d, based on increased absolute and relative
liver weights along with histopathological changes in the liver
at the LOEL of 349 mg/kg bw/d. Triflusulfuron was not oncogenic
in the mouse. The NOEL for chronic (2-yr) systemic toxicity and
oncogenicity in rats was 4.06 mg/kg bw/d, based on lower
body weight and erythrocyte counts,
and an increased incidence of Leydig cell
tumours (adenomas) in males at 30.6 mg/kg bw/d. A study
was performed that attempted to determine the mechanism of tumour
formation. The results indicated that triflusulfuron-methyl
appears to inhibit the conversion of testosterone to estradiol
by aromatase in vitro, the in vivo results are inconclusive.
Disruption of the hypothalmic-pituitary-
testicular (HPT) axis is a well-recognized mechanism of Leydig
cell adenoma formation in the rat by non-genotoxic compounds.
Aromatase inhibition is one of the established mechanisms of disruption.
A threshold for this effect exists; doses that do not disrupt
the HPT axis should not cause tumours. The
relevance of these tumours to humans is questionable, as they
are extremely rare in humans.
Ref: Dec 3, 1999 - Report on Triflusulfuron
methyl. Regulatory Note REG99-03. Pest Management Regulatory Agency,
Health Canada, Ottawa.
http://www.fluorideaction.org/pesticides/triflusulfuron.methy.canada.pdf
Gastrointestinal
(click
on for all fluorinated pesticides)
Groups of 20 artificially inseminated female rabbits were treated
by gavage with 0, 15, 90, 270 or 800 mg/kg bw/d DPX-66037-24 (95.6%
purity) on days 7Ð19 of gestation. The NOEL for maternal toxicity
was 15 mg/kg bw/d. At 90 mg/kg bw, body-weight gains were lower
than controls at the beginning of the treatment period. Clinical
signs of toxicity were observed at 270 mg/kg bw/d (stool absent
or reduced, small stool), abortions and evidence of
gastrointestinal effects were found at gross necropsy (ulceration
of gastric mucosa, gaseous distension). Food consumption
was also lower than controls in the 270 and 800 mg/kg bw/d groups...
Ref: Dec 3, 1999 - Report on Triflusulfuron
methyl. Regulatory Note REG99-03. Pest Management Regulatory Agency,
Health Canada, Ottawa.
http://www.fluorideaction.org/pesticides/triflusulfuron.methy.canada.pdf
Genotoxicity
(click
on for all fluorinated pesticides)
-- A human lymphocyte cytogenetic assay was performed using 0.5,
1.5, 1.7, 1.85, or 2.0 mg/mL DPX-66037-59 (98.7%) with and without
metabolic activation. Cytotoxicity was observed at 1.85 mg/mL,
indicated by a lower mitotic index than controls. Triflusulfuron-methyl,
with metabolic activation, was clastogenic
at 2.0 mg/mL under the conditions of this test.
-- A human lymphocyte cytogenetic assay was performed using 0.1Ð2.0
mg/mL DPX-66037-24 (95.6%) with and without metabolic activation.
Cytotoxicity was observed at 2.0 mg/mL,
indicated by a lower mitotic index than controls. Triflusulfuron-methyl,
with metabolic activation, was clastogenic at $1.7 mg/mL in a
dose-dependent manner under the conditions of this test.
-- ... It was genotoxic in mammalian in
vitro chromosomal aberrations assays (same assay performed twice),
but not in a mammalian in vivo assay.
Ref: Dec 3, 1999 - Report on Triflusulfuron
methyl. Regulatory Note REG99-03. Pest Management Regulatory Agency,
Health Canada, Ottawa.
http://www.fluorideaction.org/pesticides/triflusulfuron.methy.canada.pdf
Liver
(click
on for all fluorinated pesticides)
-- 90-Day oral toxicity
in nonrodents. NOAEL = 3.9/3.7 (m/f)
mg/kg/day LOAEL = 146.9/159.9 (m/f) mg/kg/day based on decreased
mean body weight and body weight gain, decreased hematocrit, hemoglobin,
RBC`s, SGOT, SGPT, ALP, absolute and relative liver and testes
weight; microscopic abnormalities
of the liver and testes.
-- Carcinogenicity mice NOAEL = 14.6
mg/kg/day LOAEL = 349 mg/kg/day based on increased
liver weight and increased hepatic cell tumors (adenomas and/or
carcinomas combined. (Possible) evidence of carcinogenicity
Ref: Federal Register: June 12, 2002 (Volume
67, Number 113). Triflusulfuron Methyl; Pesticide Tolerance. Final
Rule.
http://www.fluorideaction.org/pesticides/triflusulfuron.m.fr.june.02.htm
-- Chronic toxicity dogs NOAEL = 26.9 mg/kg/day LOAEL = 116.6
mg/kg/day based on increased liver weight, alkaline phosphatase,
and hepatocellular hypertrophy.
Ref: Federal Register: June 12, 2002 (Volume
67, Number 113). Triflusulfuron Methyl; Pesticide Tolerance. Final
Rule.
http://www.fluorideaction.org/pesticides/triflusulfuron.m.fr.june.02.htm
In another 90-day subchronic
study, dogs were fed dosages of 3.87, 146.1, or 267.6 mg/kg/day
(males) or 3.72, 159.9, or 250.7 mg/kg/day (females). Triflusulfuron
methyl was found to be hepatotoxic
at 4,000 ppm (146.1 mg/kg/day males and 159.9 mg/kg/day females),
and greater elevated hepatic enzyme levels and postmortem evidence,
including elevation in liver weights
and microscopic evidence of bile stasis. Other microscopic findings
considered to be treatment related were testicular atrophy and
decreased testicular weights and hypercellularity of the sternal
and femoral bone marrow, with a corresponding increase in reticulocyte
and leukocyte counts seen in the high-dose males and females.
Based on the microscopic findings in the liver
and testes of the 4,000 ppm and greater treated animals, the NOAEL
was 3.87 mg/kg/day (males) and 3.72 mg/kg/day (females).
Ref: Federal Register. August 8, 2001. [PF-1036;
FRL-6795-4]
http://www.fluoridealert.org/pesticides/Triflusulfuron.M.FR.Aug8.01.htm
Sciatic
Nerve (click
on for all fluorinated pesticides)
Groups of 62 rats/sex were fed diets containing 0, 10, 100, 750
or 1500 ppm DPX-66037- 24 (95.6% purity) (equal to 0.406, 4.06,
30.6 or 64.5 mg/kg bw/d for males and 0.546, 5.47, 41.5 and 87.7
mg/kg bw/d for females) for 22 months. The NOEL for this study
was 100 ppm (4.06 mg/kg bw/d). At 750 ppm, body
weights and body-weight gains were lower than controls
in both sexes and males had lower erythrocyte counts than controls
at most time points and an increased incidence of Leydig cell
hyperplasia compared to controls. The incidence of myelin/axonal
degeneration of the sciatic nerve was increased compared
to controls in the 1500-ppm group of females (25/48
versus [vs] 42/49)...
Ref: Dec 3, 1999 - Report on Triflusulfuron
methyl. Regulatory Note REG99-03. Pest Management Regulatory Agency,
Health Canada, Ottawa.
http://www.fluorideaction.org/pesticides/triflusulfuron.methy.canada.pdf
Teratogenic
(click
on for all fluorinated pesticides)
TERATOLOGY, RAT. 51974-030; 119842;
"Teratogenic Study of DPX-66037-24 in Rats" (Author:
C.A. Mebus; E.I. du Pont de Nemours & Company, Inc., Newark,
DE, Project No. HLR 525-91, 11/1/91); DPX-66037-24 (95.6% Triflusulfuron-methyl);
0, 30, 120, 350, 1000 mg/kg/day oral gavage; 25 Crl: CD BR female
rats/dose; observations- maternal effects; five animals died due
to dosing injuries; significant decreases in maternal weight changes
and feed consumption were observed; fetal effects; significantly
increased average number of malformed fetuses were observed,
the majority of fetal malformations occurred in one fetus, when
individual end points of developmental evaluation criteria were
grouped a significant increase was noted
in the 350 and 1000 mg/kg dose groups primarily due to retarded
renal development and partial ossification of skulls, sternebra
or vertebra; no adverse effect; Maternal NOEL = 120 mg/kg
(based on decreased maternal weight gain and feed consumption),
Developmental NOEL = 120 mg/kg (based on
increased average number of malformed fetuses, retarded fetal
development).
|

