| Reports
available from
The National Technical Information Service
(NTIS)
Order from NTIS by: phone at 1-800-553-NTIS (U.S. customers);
(703)605-6000 (other countries); fax at (703)605-6900;
and email at orders@ntis.gov. NTIS is located at 5285
Port Royal Road, Springfield, VA, 22161, USA.
•
Note from EC:
-- FAN would like to obtain these
studies. If you have copies and are willing to share,
FAN will reimburse postage and reasonable copying fees.
Or send your copy to us and we will copy and return.
-- These
studies were cited on Toxline at Toxnet in December 2004.
|
| Order
No |
Title |
Abstract
/ Keywords |
NTIS/03330068
6p |
2004
- Saber insecticide ear tags.
Pest
Management Regulatory Agency. Alternative Strategies &
Regulatory Affairs Division, Ottawa (Ontario).
Regulatory decision document no. RDD2004-02.
Text in English and French (Bilingual). French ed. on
the same fiche.
|
This
document presents the regulatory decision concerning Saber
Insecticide Ear Tags, containing the active ingredient lambda-cyhalothrin.
This product is used for the control of horn and face flies
on beef and non-lactating dairy cattle. |
NTIS/02070140
66p
• Available
for free online |
2004
- Lambda-cyhalothrin Saber Insecticide
Ear Tags.
Pest
Management Regulatory Agency. Alternative Strategies &
Regulatory Affairs Division, Ottawa (Ontario).
Proposed regulatory decision document no. PRDD2004-02.
Text in English and French (Bilingual). French ed. on
the same fiche. |
The
end-use product Saber Insecticide Ear Tags, containing lambda-cyhalothrin,
is used for the control of horn and face flies on beef and
non-lactating dairy cattle. This document provides a summary
of data reviewed and the rationale for the proposed regulatory
decision for this product. It includes information on the
following: The active substance, its properties and uses;
methods of analysis; impact on human and animal health;
residues; efficacy. |
NTIS/02170171
82p
•
Available
for free online (English) |
2003
- Lambda-cyhalothrin Demand CS
insecticide.
Pest
Management Regulatory Agency. Alternative Strategies &
Regulatory Affairs Division, Ottawa (Ontario).
Proposed regulatory decision document no. PRDD2003-03.
Text in English and French (Bilingual). French ed. on
the same fiche. |
The
end-use product Demand CS, containing the insecticide active
ingredient lambda-cyhalothrin, is proposed for the control
of structural pests (cockroaches, ants, carpenter ants)
as a perimeter treatment around buildings and as a crack
and crevice treatment in non-residential buildings and non-passenger
areas of transport vehicles. This document contains a summary
of data reviewed and the rationale for the proposed regulatory
decision. It includes information on the following: Properties
and uses of active substance; methods of analysis; impact
on human and animal health; fate and behaviour in the environment;
effects on non-target species; efficacy. |
NTIS/03360172
53p |
2001
- Lambda-cyhalothrin Saber Insecticide
Ear Tags.
Pest
Management Regulatory Agency. Submission Management &
Information Division, Ottawa (Ontario).
Regulatory note no. REG2001-04.
Text in English and French (Bilingual). French ed. on
the same fiche. |
This
note summarizes data reviewed and the rationale for the
regulatory decision on the end-use product Saber Insecticide
Ear Tag, containing lambda-cyhalothrin, used for the control
of flies on cattle. Information is included on the active
substance, its properties, & uses; methods for analysis
of the active substance, its formulation, & residues;
impacts on human & animal health arising from exposures;
residues in animal parts; effectiveness & the development
of insecticide resistance; and considerations related to
the Toxic Substances Management Policy. |
NTIS/OTS0545462
EPA/OTS;
Doc #88-920007123 |
1992
- INITIAL SUBMISSION: ACUTE ORAL AND ACUTE DERMAL TOXICITY
STUDIES WITH PP321 IN RATS WITH COVER LETTER DATED 08-28-92
IMPERIAL
CHEM INDUS PLC |
ICI
AMERS INC
PP321
HEALTH EFFECTS
ACUTE TOXICITY
MAMMALS
RATS
ORAL
GAVAGE
DERMAL
CAS
No.68085-85-8 |
NTIS/OTS0545463
EPA/OTS;
Doc #88-920007124 |
1992
- INITIAL SUBMISSION: ACUTE ORAL TOXICITYDATED 08-28-92
IMPERIAL
CHEM INDUS PLC |
ICI
AMERS INC
PP321
HEALTH EFFECTS
ACUTE TOXICITY
MAMMALS
MICE
ORAL
GAVAGE
CAS No.68085-85-8 |
NTIS/OTS0545653
EPA/OTS;
Doc #88-920007400 |
1992
- INITIAL SUBMISSION: PP321: 4-HOUR ACUTE INHALATION TOXICITY
STUDY IN THE RAT WITH ATTACHMENT AND COVER LETTER DATED
08-28-92
IMPERIAL
CHEM INDUS
|
ICI
AMERS INC
PP321
HEALTH EFFECTS
ACUTE TOXICITY
MAMMALS
RATS
INHALATION
CAS No.68085-85-8 |
NTIS/OTS0545310
EPA/OTS;
Doc #88-920006863 |
1992
- INITIAL SUBMISSION: PP321: 1 YEAR ORAL DOSING STUDY IN
DOGS WITH COVER LETTER DATED 08-28-92
|
ICI
AMERS INC
PP321
HEALTH EFFECTS
CHRONIC TOXICITY
COMBINED CHRONIC TOXICITY/CARCINOGENICITY
MAMMALS
DOGS
ORAL
GAVAGE
CAS No.68085-85-8 |
NTIS/OTS0538576
EPA/OTS;
Doc #88-920007495
|
1992
- INITIAL SUBMISSION: 4-HOUR ACUTE INHALATION TOXICITY
STUDY IN THE RAT OF A 13% EC FORMULATION WITH COVER LETTER
DATED 08-28-92
IMPERIAL
CHEM INDUS LTD |
ICI
AMERICAS INC.
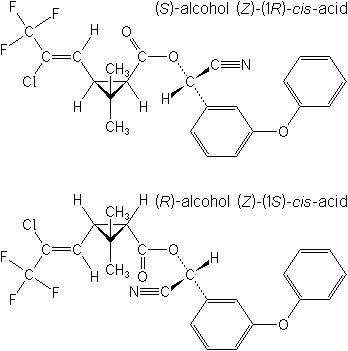
(R+S)-A-CYANO-3-PHENOXYBENZYL-(Z)
-(IRSK3RS)-3-(2-CHLORO-3,3*
HEALTH EFFECTS
ACUTE TOXICITY
MAMMALS
RATS
INHALATION
CAS No.68085-85-8 |