|
Return
to Hydramethylnon Index Page
Activity:
Insecticide
(unclassified)
Structure:
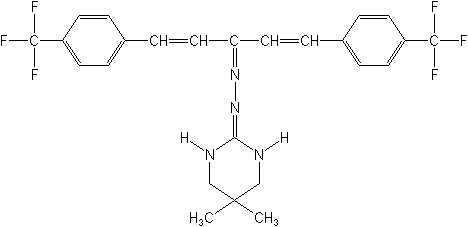
Adverse
Effects:
Amyloidosis
- Kidney
Blood
Body
Weight Decrease
(including cachexia and anorexia)
Bone
Cancer: Possible Human Carcinogen - LUNG
Endocrine:
Adrenal
Endocrine:
Suspected Endocrine Disruptor
Endocrine:
Testicular
Endocrine: Thymus
Endocrine: Uterus
Kidney
Lung
Reproductive/Developmental
Environmental
•
US: As of February 15, 2005, this herbicide is permitted
in or on
3 food commodities in the United States - see list
at bottom of page.
•
European Commission: Not allowed to be used after July 25,
2003
|
Amyloidosis
(click on for all fluorinated
pesticides)
-- In a carcinogenicity study, MRID 00101563, groups of 50 male
and 50 female Charles River CD-1 mice received diets containing
hydramethylnon at dose levels of 0, 25, 50, 100, or 200 ppm (0,
3.57, 6.93, 14.2, or 28.6 mg/kg/day in males, and 0, 4.45, 6.87,
17.3, or 33.1 mg/kg/day in females, based on food consumption)
for 18 months. The 200 ppm males and females were sacrificed after
55 weeks because of high mortality. Survival after 18 months at
the 50 and 100 ppm doses was 72% and 46% in males, and 66% and
46% in females (compared to control survival of 86% in males and
76% in females)... Dose-related amyloidosis
was seen in the kidneys of the 50 and 100 ppm females.
Ref: US EPA. Reregistration Eligibility
Decision (RED) Hydramethylnon. EPA 738-R-98-023. December 1998.
http://www.fluoridealert.org/pesticides/Hydramethylnon.RED.1998.pdf
18-Month Feeding (oncogenic)
- mouse: Systemic NOEL=25 ppm (2.75 mg/kg/day); Systemic LEL=50
ppm (3.75 mg/kg/day) (increased testicular lesions, decreased
body weight gain, increased renal amyloidosis);
core grade minimum (American Cyanamid, 1982d).
Ref: US EPA IRIS for Amdro (CASRN
67485-29-4).
http://www.epa.gov/iris/subst/0207.htm
Chronic toxicity...
In an 18-month cancer assay, hydramethylnon at about 3.8 mg/kg/day
was associated with amyloidosis, a syndrome
in which abnormal protein deposition in the kidney fitration unit
(glomerulus) results in damage [13].
Ref:
E X T O X N E T Extension Toxicology Network Pesticide Information
Profiles for Hydramethylnon.
http://ace.ace.orst.edu/info/extoxnet/pips/hydramet.htm
Blood
(click on for all fluorinated
pesticides)
PubMed abstract: Holstein
calves (3 to 5 months of age) were used to develop an animal model
sensitive to environmental toxicants. In the present study, the
fire ant toxicant AMDRO was fed (113.5
g/day/calf) to weanling castrated calves (9 test and 9 controls)
for 7 weeks. As early as 14 days after the start of the AMDRO
feeding, leukopenia was observed.
Differential counts revealed significant non-transient decreases
in lymphocytes and eosinophils. Eosinopenia
was observed from days 21 to 49 of AMDRO treatment. Variability
in hematocrit and hemoglobin values in treated and control calves
precluded making a determination of trends due to toxicant exposure...
Ref: Am J Vet Res 1984 May;45(5):1023-7;
Hematologic
and immunologic responses of Holstein calves to a fire ant toxicant,
by Evans DL, Jacobsen KL, Miller DM.
In a 21-day dermal
toxicity study in rabbits, MRID 00101559, groups of 10 male and
10 female New Zealand White rabbits received a total of 15 repeated
dermal applications of hydramethylnon at doses of 0 (control),
10, 50, or 250 mg/kg/day, 6 hours/day, 5 days/week over a three
week period... Toxicity
observed at the highest dose tested (250 mg/kg/day) included decreased
food consumption in males and females as well as thrombocytopenia
(a persistent decrease in the number of blood platelets that is
usually associated with hemorrhagic conditions) in females. Although
thrombocytopenia was observed at this dose (250 mg/kg/day), it
was not considered to be an adverse, or biologically significant
effect because it was seen in the presence of skin irritation
in animals having abraded skins. In addition, alterations in hematological
parameters are often seen in dermal toxicity studies in the presence
of skin irritation. Therefore, the 250 mg/kg/day (the highest
dose tested), in spite of the presence of this effect, is considered
to be the NOAEL for dermal and systemic toxicity; a LOAEL was
not established. MRID 00101559 is classified as acceptable and
satisfies guideline requirement 82-2 for a 21-dermal toxicity
study in rats.
Ref: US EPA. Reregistration Eligibility
Decision (RED) Hydramethylnon. EPA 738-R-98-023. December 1998.
http://www.fluoridealert.org/pesticides/Hydramethylnon.RED.1998.pdf
Body
Weight Decrease
(click
on for all fluorinated pesticides)
-- In a subchronic
toxicity study, MRID 00061794, groups of 4 male and 4 female beagles
received gelatin capsules containing hydramethylnon at doses of
0, 3.0, 6.0, or 12.0 mg/kg/day for 91 days. None of the control
or low-dose dogs died, but 3 males and 3 females in the mid-dose
died or were sacrificed moribund between days 27 and 75, and all
high-dose dogs were sacrificed moribund between days 27 and 53.
.. Body weights in the low, mid, and high-dose
groups were decreased as much as 11%, 51%, and 34% in males; and
9%, 42%, and 37% in the females (body weight decreases were greatest
in the mid-dose dogs because they survived longer than the high-dose
dogs)... All mid and high-dose dogs were
cachectic at necropsy... The 6 mg/kg/day
dose caused lethality, as well as decreased
food consumption and body weight gain, increased SGPT,
cachexia, wasting of muscle and subcutaneous
fat, and testicular atrophy. The LOAEL was 3 mg/kg/day (the lowest
does tested) based on decreased food consumption
and body weight gain; a NOAEL was not established.
-- In a 21-day dermal toxicity study in rabbits,
MRID 00101559, groups of 10 male and 10 female New Zealand White
rabbits received a total of 15 repeated dermal applications of
hydramethylnon at doses of 0 (control), 10, 50, or 250 mg/kg/day,
6 hours/day, 5 days/week over a three week period... Food consumption
was depressed as much as 38% and 45% in the high-dose males and
females, compared to controls. The high-dose
males and females weighed as much as 8% and 9% less than the controls...
Toxicity observed at the highest dose tested (250 mg/kg/day) included
decreased food consumption in males and females as well as thrombocytopenia
(a persistent decrease in the number of blood platelets that is
usually associated with hemorrhagic conditions) in females...
-- Chronic Toxicity and Carcinogenicity In a 6-month
study, MRID 00035529, groups of 4 male and 4 female beagles dogs
received gelatin capsules containing hydramethylnon at doses of
0, 0.33, 1.0, or 3.0 mg/kg/day for 26 weeks. The control group
received 120 mg/kg/day of lactose. No dogs died... A high-dose
male was removed from the study due to anorexia
between study days 42 and 98, and day 120 to termination.
-- In a chronic toxicity/carcinogenicity study, MRID 00101565,
groups of 50 male and 50 female Charles River CD rats were fed
diets containing hydramethylnon at dose levels of 0, 25, 50, 100,
or 200 ppm (0, 1.2, 2.4, 4.9, or 10.0 mg/kg/day in males, and
0, 1.5, 3.0, 6.2, or 12.1 mg/kg/day in females, respectively based
on food consumption) for two years... Body
weights in the males were as much as 17% less than the controls
at 200 ppm, and 5% at 100 ppm. Body weights in the females were
as much as 42% less than the controls at 200 ppm, and 22% at 100
ppm. Body weights were comparable in the other groups. Food consumption
was reduced an average of 7% in the 200 ppm males, and 16% in
the 200 ppm females. The other groups were comparable...
-- On May 28, 1998, the Agency's Cancer Peer Review Committee
concluded that the dose levels of 100 ppm in males, and 50 ppm
in females were adequate to assess the carcinogenic potential
of hydramethylnon in rats. This conclusion was based on significant
decreases in body weight at higher doses... Under the conditions
of this study, the NOAEL was 50 ppm (2.4 mg/kg/day in males, 3.0
mg/kg/day in females), and the LOAEL was 100 ppm (4.9 mg/kg/day
in males, 6.2 mg/kg/day in females) based on small, soft testes,
decreased testicular weights, and testicular atrophy in males;
and decreased body weight gain in females.
This study is classified as acceptable and satisfies guideline
requirement 83-5 for a chronic feeding/carcinogenicity study in
rodents.
-- In a carcinogenicity study, MRID 00101563, groups of 50 male
and 50 female Charles River CD-1 mice received diets containing
hydramethylnon at dose levels of 0, 25, 50, 100, or 200 ppm (0,
3.57, 6.93, 14.2, or 28.6 mg/kg/day in males, and 0, 4.45, 6.87,
17.3, or 33.1 mg/kg/day in females, based on food consumption)
for 18 months. The 200 ppm males and females were sacrificed after
55 weeks because of high mortality. Survival after 18 months at
the 50 and 100 ppm doses was 72% and 46% in males, and 66% and
46% in females (compared to control survival of 86% in males and
76% in females). Body weights in the 100
and 200 ppm groups were as much as 13% and 23% less than the controls
in males, and as much as 6% and 19% less than the controls in
females, respectively. Food consumption was reduced an
average of 14% in the 200 ppm males, and
20% in the 200 ppm females. The other groups were comparable...
-- In a prenatal developmental toxicity study, MRID 00061790,
groups of 26 pregnant female Sprague-Dawley rats were given oral
administration of hydramethylnon at doses of 0, 3, 10, or 30 mg/kg/day
on gestation days 6-15. The vehicle controls were dosed with corn
oil. The dams were sacrificed and examined on gestation day 20.
There were two maternal deaths in the high-dose, presumably due
to intubation error. The mid-dose dams weighed
as much as 8% less than the controls, and the high-dose dams weighed
as much as 16% less than the controls. Body weight gain
during the post-dosing interval (gestation days 15-20) was comparable
in all groups... Mean high-dose fetal weights
were reduced 10% for both sexes, but the other groups were
comparable... For maternal toxicity, the NOAEL was 3 mg/kg/day
and the LOAEL was 10 mg/kg/day, based on an 8% decrease
in body weight and yellowish discoloration of the fat...
For developmental toxicity, the NOAEL was 10 mg/kg/day and the
LOAEL was 30 mg/kg/day, based on decreased
mean fetal weights, increased incidence of rudimentary
structures, and increased incidence of incompletely ossified supraoccipital.
This study is classified as acceptable and satisfies guideline
requirement 83-3(a) for a developmental toxicity study in rats.
-- In a developmental toxicity study, MRID 00101558, groups of
16 impregnated New Zealand rabbits received oral administration
of hydramethylnon at doses of 0, 5, 10, or 20 mg/kg/day on gestation
days 6-18. The vehicle controls were dosed with corn oil. The
does were sacrificed and examined on gestation day 29. Two high-dose
does died during the post-treatment period of undetermined causes.
Six does aborted, 3 each in the mid and high-dose groups. Dose-related
clinical signs seen at the mid and high-dose included soft stool,
reduced amount of stool, and ano-genital matting and discharge.
The high-dose body weights were as much as 12% less than the controls
(gestation day 24). The low and mid-dose body weights were comparable,
though slightly less than the controls. The mean number of implantations,
corpora lutea, post-implantation loss, early or late resorptions,
viable fetuses, and sex distribution were comparable in all groups.
The fetal weights in the low, mid, and high-dose groups were 8%,
16%, and 25% lower than the controls; the low-dose was
within historical limits. For maternal toxicity, the LOAEL was
5 mg/kg/day based upon body weight established.
However, the incidence of soft stool, reduced amount of stool,
and body weight loss of less than 6%, at the low-dose, were not
considered adverse. At 10 mg/kg/day, ano-genital matting and discharge
was also observed, and the same findings, with increased severity,
were observed at the 20 mg/kg/day dose level. For developmental
toxicity, the NOAEL was 5 mg/kg/day and the LOAEL was 10 mg/kg/day,
based upon decreased fetal weight (16%)
mg/kg/day. The decreased fetal weight observed at the 5 mg/kg/day
was not considered to be treatment-related since the incidences
were within historical control ranges . This study is classified
as acceptable and satisfies guideline requirement 83-3(b) for
a developmental toxicity study in rabbits.
Ref:
US EPA. Reregistration Eligibility Decision (RED) Hydramethylnon.
EPA 738-R-98-023. December 1998.
http://www.fluoridealert.org/pesticides/Hydramethylnon.RED.1998.pdf
Bone
(click
on for all fluorinated pesticides)
Decreased fetal weight
was observed in the offspring of rats administered 30 mg/kg/day
(LOEL). The NOEL was 10 mg/kg/day. Increased post implantation
loss and decreased fetal viability were observed in the offspring
of rabbits administered 15 mg/kg/day (LOEL). The NOEL was 5 mg/kg/day.
Vertebral anomalies were seen in
the offspring of rabbits administered 10 mg/kg/day (LOEL). The
NOEL was 5 mg/kg/day.
Ref: USEPA/OPP. Support Document for the
Addition of Chemicals from Federal Insecticide, Fungicide, Rodenticide
Act (FIFRA) Active Ingredients to EPCRA Section 313. U. S. Environmental
Protection Agency, Washington, DC (1993). As cited by US EPA in:
Federal
Register: January 12, 1994. Part IV. 40 CFR Part 372. Addition
of Certain Chemicals; Toxic Chemical Release Reporting; Community
Right-to-Know; Proposed Rule.
In a prenatal developmental
toxicity study, MRID 00061790, groups of 26 pregnant female Sprague-Dawley
rats were given oral administration of hydramethylnon at doses
of 0, 3, 10, or 30 mg/kg/day on gestation days 6-15. The vehicle
controls were dosed with corn oil. The dams were sacrificed and
examined on gestation day 20... Skeletal variations were generally
comparable in all groups, although the high-dose
fetuses had an increase in the incidence of rudimentary structures
and \incompletely ossified supraoccipitals... At 30 mg/kg/day,
a 16% decrease in maternal body weight, increased incidence of
clinical signs (nasal mucus, alopecia, soft stool, staining of
the ano-genital fur), yellowish discoloration of the fat, and
small thymus were observed. For developmental toxicity, the NOAEL
was 10 mg/kg/day and the LOAEL was 30 mg/kg/day, based on decreased
mean fetal weights, increased incidence
of rudimentary structures, and increased incidence of incompletely
ossified supraoccipital. This study is classified as acceptable
and satisfies guideline requirement 83-3(a) for a developmental
toxicity study in rats.
Ref: US EPA. Reregistration Eligibility
Decision (RED) Hydramethylnon. EPA 738-R-98-023. December 1998.
http://www.fluoridealert.org/pesticides/Hydramethylnon.RED.1998.pdf
Cancer:
Possible Human Carcinogen - LUNG
(click
on for all fluorinated pesticides)
Group
C -- Possible Human Carcinogen. Lung
adenomas & combined adenomas/carcinomas; CD-1 mice (F).
Ref: April
26, 2006 . Chemicals Evaluated for Carcinogenic Potential by the
Office of Pesticide Programs. From: Jess Rowland, Chief Science
Information Management Branch Health Effect Division (7509C) Office
of Pesticide Programs, USEPA.
http://www.fluorideaction.org/pesticides/pesticides.cancer.potential.2006.pdf
Group C--Possible
Human Carcinogen. Reviewed 3/ 28/ 91.
Ref:
List of Chemicals Evaluated for Carcinogenic Potential. Science
Information Management Branch, Health Effects Division, Office
of Pesticide Programs, U. S. Environmental Protection Agency.
March 15, 2002.
http://www.biomuncie.org/chemicals_evaluated_for_carcinog.htm
The Cancer Peer Review
Committee determined that hydramethylnon should be classified
as a Group C carcinogen, a possible human carcinogen, and recommended
that, for the purpose of risk characterization, the Reference
Dose approach should be used for quantification of human risk.
This classification was based upon statistically
significant increases in lung adenomas at 50 and 100 ppm
(27% and 27%, respectively) and combined
lung adenomas/carcinomas at 25, 50, and 100 ppm (32%, 40%,
and 35%, respectively) in female mice. The MTD is between 50 ppm
and 100 ppm in both sexes of mice.
Ref: US EPA. Reregistration Eligibility
Decision (RED) Hydramethylnon. EPA 738-R-98-023. December 1998.
http://www.fluoridealert.org/pesticides/Hydramethylnon.RED.1998.pdf
Endocrine:
Adrenal
(click
on for all fluorinated pesticides)
- On May 28, 1998,
the AgencyÕs Cancer Peer Review Committee concluded that the dose
levels of 100 ppm in males, and 50 ppm in females were adequate
to assess the carcinogenic potential of hydramethylnon in rats....
The statistically significant increases
in tumors observed in the uterus
(adenomatous polyps) and adrenals
(medullary adenomas) were not considered
to be biologically significant since they were seen at excessive
doses (i.e., at 200 ppm). Under the conditions of this
study, the NOAEL was 50 ppm (2.4 mg/kg/day in males, 3.0 mg/kg/day
in females), and the LOAEL was 100 ppm (4.9 mg/kg/day in males,
6.2 mg/kg/day in females) based on small,
soft testes, decreased testicular weights, and testicular atrophy
in males; and decreased body weight gain in females. This
study is classified as acceptable and satisfies guideline requirement
83-5 for a chronic feeding/carcinogenicity study in rodents.
Ref: US EPA. Reregistration Eligibility
Decision (RED) Hydramethylnon. EPA 738-R-98-023. December 1998.
http://www.fluoridealert.org/pesticides/Hydramethylnon.RED.1998.pdf
Endocrine:
Suspected Disruptor
(click
on for all fluorinated pesticides)
"Endocrine Disruptor
Effects. EPA is required to develop a screening program to determine
whether certain substances (including all pesticides and inerts)
``may have an effect in humans that is similar to an effect produced
by a naturally occurring estrogen, or such other endocrine effect...''
The Agency is currently working with interested stakeholders,
including other government agencies, public interest groups, industry
and research scientists in developing a screening and testing
program and a priority setting scheme to implement this program.
Congress has allowed 3 years from the passage of FQPA (August
3, 1999) to implement this program. At that time,
EPA may require further testing of this active ingredient and
end use products for endocrine disruptor effects."
Ref: Federal Register. March 4, 1998. Pesticide
tolerances for emergency exemptions. Final Rule.
http://www.fluoridealert.org/pesticides/Hydramethylnon.FR.Mar4.1998.htm
Endocrine:
Testicular
(click
on for all fluorinated pesticides)
- Mutagenicity There
are five acceptable mutagenicity (84-2) studies of hydramethylnon.
The findings of adverse effects on spermatocyte
and/or spermatogonia in the dominant lethal assay are consistent
with the results of the 2-generation reproduction study in rats
showing that hydramethylnon is a reproductive
toxicant which appears to specifically target the germinal cells
and/or tissues in the testes.
- On May 28, 1998, the Agency's Cancer Peer Review Committee
concluded that the dose levels of 100 ppm in males, and 50 ppm
in females were adequate to assess the carcinogenic potential
of hydramethylnon in rats.... The statistically
significant increases in tumors observed in the uterus (adenomatous
polyps) and adrenals (medullary adenomas) were not considered
to be biologically significant since they were seen at excessive
doses (i.e., at 200 ppm). Under the conditions of this study,
the NOAEL was 50 ppm (2.4 mg/kg/day in males, 3.0 mg/kg/day in
females), and the LOAEL was 100 ppm (4.9 mg/kg/day in males, 6.2
mg/kg/day in females) based on small, soft
testes, decreased testicular weights, and testicular atrophy in
males; and decreased body weight gain in females. This
study is classified as acceptable and satisfies guideline requirement
83-5 for a chronic feeding/carcinogenicity study in rodents.
- In a carcinogenicity study, MRID 00101563, groups of 50 male
and 50 female Charles River CD-1 mice received diets containing
hydramethylnon at dose levels of 0, 25, 50, 100, or 200 ppm (0,
3.57, 6.93, 14.2, or 28.6 mg/kg/day in males, and 0, 4.45, 6.87,
17.3, or 33.1 mg/kg/day in females, based on food consumption)
for 18 months. The 200 ppm males and females were sacrificed after
55 weeks because of high mortality. Survival after 18 months at
the 50 and 100 ppm doses was 72% and 46% in males, and 66% and
46% in females (compared to control survival of 86% in males and
76% in females)... Histopathologic findings of testicular degeneration
in the 50, 100, and 200 ppm males displayed a
dose-related pattern of incidence and severity, and included hypospermia,
interstitial cell hyperplasia of Leydig cells, and germinal cell
degeneration...
- Mutagenicity There are five
acceptable mutagenicity (84-2) studies of hydramethylnon. The
findings of adverse effects on spermatocyte
and/or spermatogonia in the dominant lethal assay are consistent
with the results of the 2-generation reproduction study in rats
showing that hydramethylnon is a reproductive
toxicant which appears to specifically target the germinal cells
and/or tissues in the testes.
Ref: US EPA. Reregistration Eligibility
Decision (RED) Hydramethylnon. EPA 738-R-98-023. December 1998.
http://www.fluoridealert.org/pesticides/Hydramethylnon.RED.1998.pdf
Hydramethylnon is a
male reproductive toxicant which
appears to specifically target the germinal cells and/or tissues
in the testes... The reproductive
NOEL was 25 ppm (1.66 mg/kg/day for males) and the lowest observed
effect level (LOEL) was 50 ppm (3.32 mg/kg/day for males), based
upon histopathological findings in the testes
and the epididymides. Also, at 75 ppm (5.05 mg/kg/day in males),
reproductive performance of the males was decreased with longer
precoital intervals, lower pregnancy rates, reduced gestation
weight gain for females and smaller litters... The evidence of
male infertility and testicular
atrophy at 90 mg/kg/day in the dominant lethal assay is consistent
with similar findings observed in the chronic rat study, the 18-month
mouse feeding study, the 2-generation reproduction study, and
the 91-day oral gavage study in dogs.
Ref: Federal Register. August 14, 1998.
[PF-824; FRL-6023-2]
http://www.fluoridealert.org/pesticides/Hydramethylnon.FR.Aug.14.98.htm
In a 90-day dog feeding
study, testicular atrophy was observed
at 6 mg/ kg/day (LOEL). The NOEL was 3 mg/kg/day. In a 90-day
rat study, dietary administration of 5 mg/kg/day (LOEL) produced
testicular atrophy. The NOEL was 2.5 mg/kg/day. Dietary
administration of 6.5 mg/kg/day for 18 months produced testicular
lesions in mice. The NOEL was 2.75 mg/kg/ day. In a 2-year
rat study, dietary administration of 5 mg/kg/day produced decreased
testicular weight and testicular atrophy. The NOEL was
2.5 mg/kg/day. In a 3-generation rat reproduction study, oral
administration of 5 mg/kg/day produced male
infertility. The NOEL was 2.5 mg/kg/day... EPA
believes that there is sufficient evidence for listing hydramethylnon
on EPCRA section 313 pursuant to EPCRA section 313(d)(2)(B) based
on the available reproductive, developmental, and hepatic toxicity
data for this chemical.
Ref:
USEPA/OPP. Support Document for the Addition of Chemicals from
Federal Insecticide, Fungicide, Rodenticide Act (FIFRA) Active
Ingredients to EPCRA Section 313. U. S. Environmental Protection
Agency, Washington, DC (1993). As cited by US EPA in: Federal
Register: January 12, 1994. Part IV. 40 CFR Part 372. Addition
of Certain Chemicals; Toxic Chemical Release Reporting; Community
Right-to-Know; Proposed Rule.
Endocrine:
Thymus
(click
on for all fluorinated pesticides)
-In a prenatal developmental
toxicity study, MRID 00061790, groups of 26 pregnant female Sprague-Dawley
rats were given oral administration of hydramethylnon at doses
of 0, 3, 10, or 30 mg/kg/day on gestation days 6-15. The vehicle
controls were dosed with corn oil. The dams were sacrificed and
examined on gestation day 20... At 30 mg/kg/day, a 16% decrease
in maternal body weight, increased incidence of clinical signs
(nasal mucus, alopecia, soft stool, staining of the ano-genital
fur), yellowish discoloration of the fat, and small
thymus were observed. For developmental toxicity, the NOAEL
was 10 mg/kg/day and the LOAEL was 30 mg/kg/day, based on decreased
mean fetal weights, increased incidence of rudimentary structures,
and increased incidence of incompletely ossified supraoccipital.
This study is classified as acceptable and satisfies guideline
requirement 83-3(a) for a developmental toxicity study in rats.
Ref: US EPA. Reregistration Eligibility
Decision (RED) Hydramethylnon. EPA 738-R-98-023. December 1998.
http://www.fluoridealert.org/pesticides/Hydramethylnon.RED.1998.pdf
Endocrine:
Uterus
(click
on for all fluorinated pesticides)
- On May 28, 1998,
the Agency's Cancer Peer Review Committee concluded that the dose
levels of 100 ppm in males, and 50 ppm in females were adequate
to assess the carcinogenic potential of hydramethylnon in rats....
The statistically significant increases
in tumors observed in the uterus
(adenomatous polyps) and adrenals (medullary adenomas) were not considered to be biologically
significant since they were seen at excessive doses (i.e., at
200 ppm). Under the conditions of this study, the NOAEL was 50
ppm (2.4 mg/kg/day in males, 3.0 mg/kg/day in females), and the
LOAEL was 100 ppm (4.9 mg/kg/day in males, 6.2 mg/kg/day in females)
based on small, soft testes, decreased testicular
weights, and testicular atrophy in males; and decreased
body weight gain in females. This study is classified as acceptable
and satisfies guideline requirement 83-5 for a chronic feeding/carcinogenicity
study in rodents.
Ref: US EPA. Reregistration Eligibility
Decision (RED) Hydramethylnon. EPA 738-R-98-023. December 1998.
http://www.fluoridealert.org/pesticides/Hydramethylnon.RED.1998.pdf
Kidney
(click
on for all fluorinated pesticides)
Subchronic Toxicity
- In a 90 day feeding study in rats, MRID 00032641, groups of
20 male and 20 female Sprague-Dawley rats were dosed with hydramethylnon
in their feed at 0, 50, 100, 200, or 400 ppm (equivalent to 0,
2.5, 5.0, 10.0 or 20.0 mg/kg/day). Due to significant decreases
in body weight gain and food consumption during the first two
weeks of the study at the highest dose (400 ppm, 20 mg/kg/day),
this dose level was reduced to 25 ppm (1.25 mg/kg/day) on study
day 15. Thus, the dose levels tested were 0, 25, 50, 100, or 200
ppm (0, 1.25, 2.5, 5.0, or 10.0 mg/kg/day)... On study day 68,
a 50 ppm male was sacrificed moribund, and a 200 ppm (10.0 mg/kg/day)
female died. The 200 ppm female had a blood
urea nitrogen (BUN) value 4-fold higher than that of the controls
on day 45. Histopathologic evaluation of this female revealed
nephrocalcinosis and hydronephrosis...
-- In a carcinogenicity study, MRID 00101563, groups of 50 male
and 50 female Charles River CD-1 mice received diets containing
hydramethylnon at dose levels of 0, 25, 50, 100, or 200 ppm (0,
3.57, 6.93, 14.2, or 28.6 mg/kg/day in males, and 0, 4.45, 6.87,
17.3, or 33.1 mg/kg/day in females, based on food consumption)
for 18 months. The 200 ppm males and females were sacrificed after
55 weeks because of high mortality. Survival after 18 months at
the 50 and 100 ppm doses was 72% and 46% in males, and 66% and
46% in females (compared to control survival of 86% in males and
76% in females)... Dose-related amyloidosis
was seen in the kidneys of the 50 and 100 ppm females.
-- In a chronic toxicity/carcinogenicity study, MRID 00101565,
groups of 50 male and 50 female Charles River CD rats were fed
diets containing hydramethylnon at dose levels of 0, 25, 50, 100,
or 200 ppm (0, 1.2, 2.4, 4.9, or 10.0 mg/kg/day in males, and
0, 1.5, 3.0, 6.2, or 12.1 mg/kg/day in females, respectively based
on food consumption) for two years... Body
weights in the males were as much as 17% less than the controls
at 200 ppm, and 5% at 100 ppm. Body weights in the females were
as much as 42% less than the controls at 200 ppm, and 22% at 100
ppm. Body weights were comparable in the other groups. Food consumption
was reduced an average of 7% in the 200 ppm males, and 16% in
the 200 ppm females. The other groups were comparable...
Glomerulonephrosis was greater in the treated
males and females than in the controls, but there was no
dose-response relationship.
Ref: US EPA. Reregistration Eligibility
Decision (RED) Hydramethylnon. EPA 738-R-98-023. December 1998.
http://www.fluoridealert.org/pesticides/Hydramethylnon.RED.1998.pdf
18-Month Feeding (oncogenic)
- mouse: Systemic NOEL=25 ppm (2.75 mg/kg/day); Systemic LEL=50
ppm (3.75 mg/kg/day) (increased testicular lesions, decreased
body weight gain, increased renal amyloidosis);
core grade minimum (American Cyanamid, 1982d).
Ref: US EPA IRIS for Amdro (CASRN
67485-29-4).
http://www.epa.gov/iris/subst/0207.htm
Chronic toxicity... In an 18-month cancer assay, hydramethylnon
at about 3.8 mg/kg/day was associated with amyloidosis,
a syndrome in which abnormal protein deposition in the kidney
fitration unit (glomerulus) results in damage [13].
Ref: E X T O X N E T Extension Toxicology
Network Pesticide Information Profiles for Hydramethylnon.
http://ace.ace.orst.edu/info/extoxnet/pips/hydramet.htm
Lung
(click
on for all fluorinated pesticides)
Group
C -- Possible Human Carcinogen. Lung
adenomas & combined adenomas/carcinomas; CD-1 mice (F).
Ref: April
26, 2006 . Chemicals Evaluated for Carcinogenic Potential by the
Office of Pesticide Programs. From: Jess Rowland, Chief Science
Information Management Branch Health Effect Division (7509C) Office
of Pesticide Programs, USEPA.
http://www.fluorideaction.org/pesticides/pesticides.cancer.potential.2006.pdf
The Cancer Peer Review
Committee determined that hydramethylnon should be classified
as a Group C carcinogen, a possible human carcinogen, and recommended
that, for the purpose of risk characterization, the Reference
Dose approach should be used for quantification of human risk.
This classification was based upon statistically
significant increases in lung adenomas at 50 and 100 ppm
(27% and 27%, respectively) and combined
lung adenomas/carcinomas at 25, 50, and 100 ppm (32%, 40%,
and 35%, respectively) in female mice. The MTD is between 50 ppm
and 100 ppm in both sexes of mice.
Ref: US EPA. Reregistration Eligibility
Decision (RED) Hydramethylnon. EPA 738-R-98-023. December 1998.
http://www.fluoridealert.org/pesticides/Hydramethylnon.RED.1998.pdf
Reproductive
/ Developmental
(click
on for all fluorinated pesticides)
- Mutagenicity There
are five acceptable mutagenicity (84-2) studies of hydramethylnon.
The findings of adverse effects on spermatocyte
and/or spermatogonia in the dominant lethal assay are consistent
with the results of the 2-generation reproduction study in rats
showing that hydramethylnon is a reproductive
toxicant which appears to specifically target the germinal cells
and/or tissues in the testes.
- On May 28, 1998, the Agency's Cancer Peer Review Committee
concluded that the dose levels of 100 ppm in males, and 50 ppm
in females were adequate to assess the carcinogenic potential
of hydramethylnon in rats.... The statistically
significant increases in tumors observed in the uterus (adenomatous
polyps) and adrenals (medullary adenomas) were not considered
to be biologically significant since they were seen at excessive
doses (i.e., at 200 ppm). Under the conditions of this study,
the NOAEL was 50 ppm (2.4 mg/kg/day in males, 3.0 mg/kg/day in
females), and the LOAEL was 100 ppm (4.9 mg/kg/day in males, 6.2
mg/kg/day in females) based on small, soft
testes, decreased testicular weights, and testicular atrophy in
males; and decreased body weight gain in females. This
study is classified as acceptable and satisfies guideline requirement
83-5 for a chronic feeding/carcinogenicity study in rodents.
- In a carcinogenicity study, MRID 00101563, groups of 50 male
and 50 female Charles River CD-1 mice received diets containing
hydramethylnon at dose levels of 0, 25, 50, 100, or 200 ppm (0,
3.57, 6.93, 14.2, or 28.6 mg/kg/day in males, and 0, 4.45, 6.87,
17.3, or 33.1 mg/kg/day in females, based on food consumption)
for 18 months. The 200 ppm males and females were sacrificed after
55 weeks because of high mortality. Survival after 18 months at
the 50 and 100 ppm doses was 72% and 46% in males, and 66% and
46% in females (compared to control survival of 86% in males and
76% in females)... Histopathologic findings of testicular degeneration
in the 50, 100, and 200 ppm males displayed a
dose-related pattern of incidence and severity, and included hypospermia,
interstitial cell hyperplasia of Leydig cells, and germinal cell
degeneration...
- Mutagenicity There are five
acceptable mutagenicity (84-2) studies of hydramethylnon. The
findings of adverse effects on spermatocyte
and/or spermatogonia in the dominant lethal assay are consistent
with the results of the 2-generation reproduction study in rats
showing that hydramethylnon is a reproductive
toxicant which appears to specifically target the germinal cells
and/or tissues in the testes.
Ref: US EPA. Reregistration Eligibility
Decision (RED) Hydramethylnon. EPA 738-R-98-023. December 1998.
http://www.fluoridealert.org/pesticides/Hydramethylnon.RED.1998.pdf
| Environmental
(click on for
all fluorinated pesticides)
The
96-hour LC 50 in the Chanel Catfish was 90
ppb. Bioaccumulation factors
in bluegill sunfish are 1300
for the whole fish, 780 for
the fillet, and 1900 for viscera.
EPA believes that there is sufficient evidence for listing
hydramethylon on EPCRA section 313 pursuant to EPCRA section
313(d)(2)(C) based on the available environmental toxicity
data and the potential for bioaccumulation.
Ref: USEPA/OPP. Support
Document for the Addition of Chemicals from Federal Insecticide,
Fungicide, Rodenticide Act (FIFRA) Active Ingredients to
EPCRA Section 313. U. S. Environmental Protection Agency,
Washington, DC (1993). As cited by US EPA in: Federal
Register: January 12, 1994. Part IV. 40 CFR Part 372.
Addition of Certain Chemicals; Toxic Chemical Release Reporting;
Community Right-to-Know; Proposed Rule
Hydramethylon
Table 9: Freshwater Fish Acute Toxicity
Ref:
US EPA. Reregistration Eligibility Decision (RED) Hydramethylnon.
EPA 738-R-98-023. December 1998. http://www.fluoridealert.org/pesticides/Hydramethylnon.RED.1998.pdf |
| Species/
(Flow-through or Static) |
% ai |
96-hour
LC50 (ppm)* |
Toxicity
Category |
MRID
|
Study
Classification |
| Rainbow
trout (Oncorhynchus mykiss) |
92%
|
0.15 |
Highly toxic |
00052857
|
Acceptable |
| Bluegill
sunfish (Lepomis macrochirus) |
92% |
1.7
|
Moderately |
00061708
|
Acceptable |
| Channel
catfish (Ictalurus punctatus) |
92%
|
0.09
|
Very
highly toxic |
00061707 |
Acceptable |
|
A
February
15, 2005,
check at the Code
of Federal Regulations for Hydramethylnon: this insecticide
is permitted in or on 3
food
commodities in the United States.
The
following list identifies these crops for which EPA has
set pesticide tolerances.
|
| [Code
of Federal Regulations]
[Title 40, Volume 22]
[Revised as of July 1, 2004]
From the U.S. Government Printing Office via GPO Access
[CITE: 40CFR180.395]
[Page 428-429]
TITLE 40--PROTECTION OF ENVIRONMENT
CHAPTER I--ENVIRONMENTAL PROTECTION AGENCY (CONTINUED)
PART 180_TOLERANCES AND EXEMPTIONS FROM TOLERANCES FOR PESTICIDE
CHEMICALS
IN FOOD--Table of Contents
Subpart C_Specific Tolerances
Sec. 180.395 Hydramethylnon; tolerances for residues.
(a) General. Tolerances are established for residues of the
insecticide tetrahydro-5,5-dimethyl-2(1H)-pyrimidinone(3-(4-(tri
fluoro
methyl) phenyl)-1-(2-(4-(tri fluoro methyl) phenyl) ethenyl)-2-pro
penyl
i dene)hy dra zone in or on the following raw agricultural
commodities: |
| Commodity |
As
of
October 8,
2003
PPM |
As
of
February 15,
2005
PPM |
CFR |
Grass (pasture
and rangeland)
|
0.05 |
0.05 |
180.395 |
| Grass hay
(pasture and rangeland) |
0.05 |
0.05 |
180.395 |
| GRASS,
RANGE, HAY |
0.05 |
Not
listed this way |
180.395 |
| Pineapple |
0.05 |
0.05 |
180.395 |
| (b)
Section 18 emergency exemptions.
Time-limited tolerances are
established for residues of the insecticide hydramethylnon;
tetrahydro-
5,5-dimethyl-2-(1H)-pyrimidinoine(3-(4-trifluoromethyl)phenyl)-1-[2-
[4(trifluoromethly)phenyl]ethenyl)-2-propenylidene) hydrazone
in
connection with the use of the pesticides under section 18
emergency
exemptions granted by EPA. The tolerance will expire and is
revoked on
the date specified in the following table. |
| Commodity |
As
of
October 8,
2003
PPM |
As
of
February 15,
2005
PPM |
Expiration/
Revocation Date |
| Pineapple
|
0.05 |
0.05 |
6/30/05 |
| (c)
Tolerances with regional registrations. [Reserved]
[[Page 429]]
(d) Indirect or inadvertent residues. [Reserved] |
|

