|
Return to Adverse
Effects
ACTIVITY:
Herbicide (sulfonylurea)
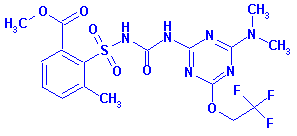
Systematic Name: Benzoic acid, 2-(((((4-(dimethylamino)-6-(2,2,2-trifluoroethoxy)-1,3,5-triazin-2-yl)amino)carbonyl)amino)sulfonyl)-3-methyl-,
methyl ester
Structure:
| Adverse
Effects:
Anemia
Blood
Body Weight Decrease
Bone
Brain
Cancer: Possible Human Carcinogen -
BLOOD, TESTES, LIVER
Endocrine: Testicular
Gastrointestinal
Genotoxicity
Liver
Sciatic nerve
Teratogenic |
| Environmental
Effects:
Potential
Ground Water Contaminant |
Regulatory
Information
(only comprehensive for the US) |
| US
EPA Registered: |
Yes |
| US
EPA PC Code: |
129002 |
| California
Chemical Code |
3875 |
| US
Tolerances: |
CFR
180.492 |
| FDA
LMS Code: |
A62 |
Registered
use in
(includes only a limited list of countries)
|
Canada,
Denmark, Finland, Germany, Netherlands, UK,
US |
US
Maximum Residue Levels permitted
in food commodities
|
Beet
(sugar, roots & tops), Chicory root |
| Other
Information |
| Molecular
Formula: |
C17-H19-F3-N6-O6-S
|
| Entry
Year: |
1990 |
| Inventing
Company: |
DuPont |
| Manufacturers: |
DuPont |
| Other
Names: |
Debut
DPX-66037
352-EUP-160
IN-66037
Pinnacle
Safari
Upbeet |
| Of
special interest: |
| PAN
Data |
| Material
Safety Data Sheets & Labels |
| Nov
18, 2005 - Summary
of Toxicology Data. California Environmntal Protection Agency.
Department of Pesticide Regulation. Medical Toxicology Branch. |
| Dec
3, 1999 - Report on Triflusulfuron
methyl. Regulatory Note REG99-03. Pest
Management Regulatory Agency, Health Canada, Ottawa. |
| October
2001 - Glossary
of Pesticide Chemicals. A listing
of pesticides subject to analysis of residues in foods and feeds
by the US Food and Drug Administration. |
| April
2000 -
Food and Drug Administration
Pesticide Residue Monitoring. - Table
3. Pesticides detectable by methods used in 1999 regulatory
monitoring. |
| Herbicide
products - partial list |
| See
also:
Triflusulfuron |
US
Federal Register
••
Note: Due to length, the following is a partial
list. Click
here to see full list of FR entries.
|
| Date
Pubilshed |
Docket
Identification Number |
Details |
| June
12, 2002 |
OPP-2002-0082
|
IR-4
* and Dupont.
Pesticide Tolerance
for residues in or on Beet (sugar roots and tops) and Chicory
(roots) -- all at 0.05 ppm. FINAL RULE.
-- Chronic toxicity rodents
NOAEL = 2.44 mg/kg/day LOAEL = 30.6 mg/kg/day based on decreased
body weight and body weight gain, alteration in hematology (mainly
males) and increased incidences of interstitial
cell hyperplasia in testes.
-- Chronic toxicity dogs
NOAEL = 26.9 mg/kg/day LOAEL = 116.6 mg/kg/day based on increased
liver weight, alkaline phosphatase,
and hepatocellular hypertrophy.
-- Carcino-genicity rats
NOAEL = 2.44 mg/kg/day LOAEL = 30.6 mg/kg/day based on decreased
body weight and body weight gain, alteration in hematology
(mainly males) and increased incidences
of interstitial cell hyperplasia in the testes. (Possible)
evidence of carcinogenicity
-- Carcino-genicity mice NOAEL
= 14.6 mg/kg/day LOAEL = 349 mg/kg/day based on increased
liver weight and increased hepatic
cell tumors (adenomas and/or carcinomas combined. (Possible)
evidence of carcinogenicity.
-- Special studies: In vivo The purpose
of these studies was to and in vitro mechanic investigate the
mechanism of Leydig cell studies
tumor induction in the testes of male rats.
A dose-dependent decrease in aromatase enzyme activity was seen
in vitro, but was inconclusive
in vivo. |
| August
8, 2001 |
PF-1036 |
IR-4
* - Pesticide
Tolerance Petition for residues of triflusulfuron methyl in
or on the raw agricultural commodity chicory (root) at 0.05
ppm. Additional data may be needed before EPA rules on
the petition. This notice includes a summary of the petition
prepared by Dupont, E.I. du Pont Nemours and Company. Comments
due by September 7, 2001. |
| Dec
22, 1999 |
PF-899 |
Dupont
- Petition
to Establish a Tolerance for residues in or on the raw agricultural
commodity [beet, sugar, root and beet, sugar, top at 0.05
ppm. |
|