|
Return to
Index Page
Adverse
Effects
ACTIVITY: Fungicide
(Strobin)
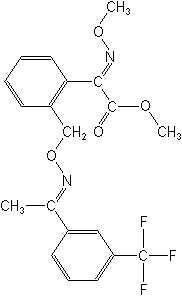
CAS Name: methyl
(?E)-?-(methoxyimino)-2-[[[(E)-[1-[3-(trifluoromethyl)phenyl]ethylidene]amino]oxy]methyl]benzeneacetate
Structure:
| |
| Date
Published |
Docket
Identification Number |
Details |
September 19, 2007
|
EPA-HQ-OPP-2007-0539
|
Bayer CropScience. Pesticide tolerance. FINAL RULE. This regulation establishes a tolerance for combined residues of Trifloxystrobin and the acid metabolite CGA-321113, at higher levels than requested by Bayer, in or on
-- Grass, forage at 12 ppm (Bayer requested 10.0 ppm)
-- Grass, hay at 17 ppm (Bayer requested 14.0 ppm)
April 11, 2007: Human Health Risk Assessment for Section 3 Registration for the Proposed Uses on Grasses Grown for Seed.
Based upon review of the data supporting the petition, EPA has modified the tolerances proposed for grass, forage and grass, hay. The appropriate tolerances for grass forage and hay were calculated to be 12 and 17 ppm, respectively. Although residue data were also provided for grass straw and seed screenings, tolerances are not required on these commodities as the Agency no longer considers them to be significant livestock feedstuffs. The recommended tolerance levels for grass forage and hay were determined considering recent Agency Guidance (Guidance for Setting Pesticide Tolerances Based on Field Trial Data).
Conclusion. EPA has determined that reliable data show that it would be safe for infants and children to reduce the FQPA safety factor to 1X. That decision is based on the following findings:
i. The toxicity database for Trifloxystrobin is complete except for an acute neurotoxicity study which is classified as unacceptable. The toxicity database contains developmental toxicity studies in two species (rats and rabbits) and a 2-generation reproduction study in rats which are adequate to assess pre and/or post natal susceptibility to infants and children. Although the available, submitted acute neurotoxicity study was found to be unacceptable, based on a weight-of- the evidence review of the available data, the lack of this study does not impact the Agency's ability to make an FQPA safety factor decision. Given that there was no evidence of neurotoxicity in this study at the Limit Dose nor in the other subchronic and chronic studies in the database there is not an uncertainty concerning neurotoxic effects and EPA has reliable data to show that removal of the FQPA safety factor is safe for children. Additionally, these data demonstrate that a developmental neurotoxicity study is not required for this pesticide. |
| August 1, 2007 |
EPA-HQ-OPP-2007-0539 |
Bayer CropScience.
Pesticide
Petition PP 6F7024. Proposal to establish a tolerance
for residues of the fungicide trifloxystrobin in or on food
commodities
-- grass, forage at 10.0 ppm
-- grass, hay at 14.0 ppm.
A practical analytical methodology for detecting
and measuring levels of trifloxystrobin in or on raw agricultural
commodities has been submitted. The limit of detection (LOD)
for each analyte of this method is 0.08 ng injected, and the
limit of quantitation (LOQ) is 0.02 ppm. The method is based
on crop specific cleanup procedures and determination by gas
chromatography with nitrogen-phosphorus detection. |
| September 22, 2006 |
EPA-HQ-OPP-2005-0299 |
Bayer CropScience.
Pesticide
Tolerance. FINAL
RULE.
in or on
-- soybean, forage at 10.0 ppm
-- soybean, hay at 25.0 ppm
-- soybean, seed at 0.08 ppm.
|
| September 13, 2006 |
EPA-HQ-OPP-2006-0730 |
IR-4.
Pesticide
petition: PP 6E7088.
Proposing the establishment of new tolerances in or on
-- asparagus at 0.07 ppm
-- papaya at 0.4 ppm
-- sapote, black at 0.4 ppm
-- canistel at 0.4 ppm
-- sapote, mamey at 0.4 ppm
-- mango at 0.4 ppm
-- sapodilla at 0.4 ppm
-- star apple at 0.4 ppm
-- radish, tops at 20 ppm
-- vegetable, root, except sugar beet, subgroup
1B at 0.2 ppm
This subgroup includes 19
commodities.
beet, garden • burdock, edible • carrot •
carrot culls • celeriac • chervil, turnip rooted
• chicory root • ginseng • horseradish
• parsley, turnip rooted • parsnip • radish
• radish, oriental • rutabaga • salsify
• salsify, black • salsify, spanish •
skirret • turnip
|
| June 28, 2006 |
EPA-HQ-OPP-2006-0084 |
Notice
of Receipt of Requests to Voluntarily Cancel Certain Pesticide
Registrations.
| EPA Registration No. |
Product Name |
Registrant |
| 000264 WA-01-003 |
Stratego Fungicide |
Bayer Cropscience LP
2 T.W. Alexander Drive
Research Triangle Park, NC 27709. |
|
| March
29, 2006 |
EPA-HQ-OPP-2005-0299 |
Bayer
CropScience and IR-4.
Pesticide Tolerance.
FINAL RULE.
This
regulation establishes a tolerance for combined residues of
trifloxystrobin and the free form of its
acid metabolite CGA-321113 ((E,E)-methoxyimino-[2-[1-(3-trifluoromethylphenyl)-ethylideneaminooxymethyl]-
phenyl]acetic acid) pesticide petition (PP 4F6892) in or on
| Commodity
|
Parts
per million |
Registrant |
Pesticide
Petition |
| Barley,
grain |
0.05 |
IR-4 |
PP
3E6769 |
| Barley,
hay |
0.3 |
IR-4 |
PP
3E6769 |
| Barley,
straw |
5.0 |
IR-4 |
PP
3E6769 |
Corn,
sweet, cannery waste
|
0.6 |
BAYER |
PP
4F6892 |
| Corn,
sweet, forage |
0.6 |
BAYER |
PP
4F6892 |
| Corn,
sweet, kernel plus cob with husks removed |
0.04 |
BAYER |
PP
4F6892 |
| Corn,
sweet, stover |
0.25 |
BAYER |
PP
4F6892 |
| Oat,
forage |
0.3 |
IR-4 |
PP
3E6769 |
| Oat,
grain |
0.05 |
IR-4 |
PP
3E6769 |
| Oat,
hay |
0.3 |
IR-4 |
PP
3E6769 |
| Oat,
straw |
5.0 |
IR-4 |
PP
3E6769 |
•
Specific information on the studies received and the nature
of the toxic effects caused by trifloxystrobin and CGA-321113
as well as the no-observed adverse effect level (NOAEL) and
the lowest observed adverse effect level (LOAEL) from the
toxicity studies can be found in the Federal
Register of May 22, 2002
•
Effects cited only from rat studies:
decreases in body weight, body weight gains, reduced
food consumption and histopathological lesions in the
liver, kidneys and spleen
•
Cancer.
EPA determined that trifloxystrobin should be classified as
a ``Not Likely Human Carcinogen.'' Due
to the classification, no cancer exposure assessment was performed.
•
Conclusion.
EPA determined that the 10X SF to protect infants and children
should be reduced to 1X. The
FQPA, SF is reduced because ...
iii. EPA determined that a developmental
neurotoxicity study in rats is not required;
iv. Although an acute neurotoxicity
study is required (the submitted study was unacceptable),
the lack of an acute neurotoxicity study does not impact EPA's
ability to make an FQPA safety factor decision because upgrading
the study would not result in a lower NOAEL than what is present
for the acute RfD ... |
| Jan
18, 2006 |
EPA-HQ-OPP-
2005-0546 |
IR-4.
Amendment
to Existing Tolerance. PP 3E6769.
IR-4, 681 U.S. Highway #1 South, North Brunswick, NJ 08902,
proposes to amend 40 CFR 180.555 by establishing tolerances
for the combined residues of the fungicide
trifloxystrobin and the free form of its acid metabolite (CGA-32113)
in or on the following food commodities. A practical analytical
methodology for detecting and measuring levels of trifloxystrobin
in or on food commodities has been submitted to the EPA. The
limit of detection (LOD) for each analyte of this method is
0.08ng injected, and the limit of quantitation (LOQ) is 0.02
ppm. The method is based on crop specific cleanup procedures
and determination by gas chromatography with nitrogen-phosphorus
detection. A description of the analytical method available
for the detection and measurement of the pesticide chemical
residues is available on EPA's Electronic Docket at http://www.regulations.gov/.
| Commodity |
PPM |
| barley,
grain |
0.05 |
| barley,
hay |
0.3 |
| barley,
straw |
5.0 |
| oat,
forage |
0.3 |
|
oat, grain |
0.05 |
| oat,
hay |
0.3 |
| oat,
straw |
5.0 |
|
| Jan
4, 2006 |
EPA-HQ-OPP-2005-0299 |
Bayer
CropScience.
Pesticide
Petitions for New Tolerances for Residues of Trifloxystrobin
in or on Corn and Soybeans.
Bayer
CropScience,
P.O. Box 12014, 2 T.W. Alexander Drive, Research Triangle
Park, NC 27709, proposes the following new tolerances. The
limit of detection (LOD) for each analyte of this method is
0.08 ng injected, and the limit of quantitation (LOQ) is 0.02
ppm. The method is based on crop-specific cleanup procedures
and determination by gas chromatography with nitrogen-phosphorus
detection.
| Pesticide
Petitions |
Commodity |
PPM |
| PP
4F6892 |
corn,
sweet (kernel plus cob with husks removed) |
0.04 |
| PP
4F6892 |
corn,
sweet, forage |
0.6 |
| PP
4F6892 |
corn,
sweet, stover |
0.25 |
| PP
5F6956 |
soybean,
seed |
0.08 |
| PP
5F6956 |
soybean,
forage |
8.0
|
| PP
5F6956 |
soybean,
hay |
20.0
|
| PP
5F6956 |
soybean,
grain aspirated fractions |
4.2
|
|
| August
31, 2005 |
OPP-2005-0223 |
Pesticide
Emergency Exemptions:
•
Colorado.
EPA authorized the use of trifloxystrobin
on soybeans to control soybean
rust; April 8, 2005 to November 10, 2007. Contact: (Andrew
Ertman).
• Georgia. Quarantine:
EPA authorized the use of trifloxystrobin
on soybeans to
control soybean rust; April 8, 2005 to November 10, 2007.
Contact: (Andrew Ertman).
• New Jersey. Quarantine:
EPA authorized the use of trifloxystrobin
on soybeans to
control soybean rust; April 8, 2005 to November 10, 2007.
Contact: (Andrew Ertman).
• Tennessee. Quarantine:
EPA authorized the use of trifloxystrobin
on soybeans to
control soybean rust; April 8, 2005 to November 10, 2007.
Contact: (Andrew Ertman).
• Vermont. Quarantine:
EPA authorized the use of trifloxystrobin
on soybeans to
control soybean rust; June 23, 2005 to November 10, 2007.
Contact: (Andrew Ertman).
• West Virginia. EPA authorized
the use of trifloxystrobin on
soybeans to control
soybean rust; April 8, 2005 to November 10, 2007. Contact:
(Andrew Ertman).
• Wisconsin. EPA authorized
the use of trifloxystrobin on
soybeans to control
soybean rust; April 8, 2005 to November 10, 2007. Contact:
(Andrew Ertman). |
| August
3, 2005 |
OPP-2005-0201 |
Cancellation
of Pesticides for Non-payment of Year 2005 Registration Maintenance
Fees.
| Section
24(c) Registrations canceled for non-payment of the
2005
maintenance fee are shown in the following Table 1:
Table
1.--Section 24(c) Registrations Canceled for Non-Payment
of Maintenance Fee |
|
SLN no. |
Product
Name |
| 000264
ND-01-0003 |
Stratego
Fungicide |
|
| June
24, 2005 |
OPP-2005-0155 |
Pesticide
Tolerances for Emergency Exemptions. FINAL
RULE.
Time-limited tolerances are established for combined residues
of the fungicide trifloxystrobin and the free form of its acid
metabolite CGA-321113 ((E,E)-methoxyimino-[2-[1-(3-trifluoromethylphenyl)
ethylideneaminooxymethyl]phenyl]acetic acid):
| Commodity |
Parts
per million |
Expiration/
revocation date |
| Soybean,
forage |
4.0
|
12/31/09 |
| Soybean,
hay |
6.5
|
12/31/09 |
| Soybean,
seed |
0.04 |
12/31/09 |
| Exposure/scenario |
Study
and toxicological effects |
Acute
dietary
(Females 13-49 years of age) |
Developmental
toxicity-- rat
LOAEL = 500 mg/kg/day based on increased
fetal skeletal anomalies. |
Chronic
dietary
(All populations) |
2-Generation
reproduction study-- rat
LOAEL = 55.3 mg/kg/day based on decreases
in body weight, body weight gains, reduced food
consumption, and histopathological
lesions in the liver, kidneys, and spleen. |
Short-
(1 to 30 days) and intermediate-term (1-6 months)
Oral |
2-Generation
reproduction study-- rat
LOAEL = 55.3 mg/kg/day based on reduced
pup body weights during lactation. |
Short-
(1 to 30 days) and intermediate-term (1-6 months)
Dermal |
28-Day
dermal toxicity study--rat
LOAEL = 1,000 mg/kg/day based on increases in mean absolute
and relative liver and kidney
weights. |
Long-term
(> 6 months)
Dermal |
2-Generation
reproduction study-- rat
LOAEL = 55.3 mg/kg/day based on decreases
in body weight, body weight gains, reduced food
consumption, and histopathological
lesions in the liver, kidneys, and spleen. |
Short-
(1 to 30 days), intermediate- (1-6 months), and long-term
(> 6 months)
Inhalation |
2-Generation
reproduction study-- rat
LOAEL = 55.3 mg/kg/day based on decreases
in body weight, body weight gains, reduced food
consumption, and histopathological
lesions in the liver, kidneys, and spleen. |
Cancer
(Oral, dermal, and inhalation) |
Classification:
``Not Likely Human Carcinogen,'' based on the lack of
evidence of carcinogenicity in mouse and rat cancer studies.
["Accordingly,
no additional cancer risk assessment was performed for
trifloxystrobin."] |
|
| Feb
23, 2005 |
OPP-2005-0037 |
Pesticide
Emergency Exemptions.
• Minnesota Department of
Agriculture
Quarantine: EPA authorized the use of trifloxystrobin on soybeans
to control soybean rust; December 13, 2004, to December 1, 2007.
• South Dakota Department
of Agriculture
Quarantine: EPA authorized the use of trifloxystrobin on soybeans
to control soybean rust; December 13, 2004, to December 1, 2007.
|
| Oct
27, 2004 |
OPP-2004-0338 |
Cancellation
of Pesticides for Non-payment of Year 2004 Registration Maintenance
Fees.
Table
1.--Section 24(c) Registrations Canceled for Non-Payment
of
Maintenance Fee |
| Name
of Pesticide |
Product
Name |
Registrant |
SLN
No. |
| Trifloxystrobin |
Stratego
Fungicide |
Bayer
Cropscience Lp |
003125
OR-03-0005 |
|
| Sept
10, 2003 |
OPP-2003-0244 |
Pesticide
Tolerance. FINAL
RULE.
--
Trifloxystrobin is currently registered for use on the following
residential non-dietary sites: Turfgrass and ornamental (CompassTM).
-- EPA determined that a developmental neurotoxicity study
in rats is not required.
-- Although
an acute neurotoxicity study is required (the
submitted study was unacceptable), the lack of an acute
neurotoxicity study does not impact EPA's ability to make
an FQPA SF decision.
-- For the toxic effects caused by trifloxystrobin EPA directs
you to the May 22, 2002, Federal Registe.
-- Developmental
toxicity-
Rat. LOAEL = 500 mg/kg/day,. based upon
increased fetal skeletal anomalies
-- 2-generation reproduction study-Rat.
LOAEL = 55.3 mg/kg/day, based upon decreases
in body weight, body weight gains, reduced food consumption
and histopathological lesions in the liver, kidneys and spleen
and pup
body weights during lactation
-- 28-Day dermal toxicity study-Rat.
LOAEL = 1,000 mg/kg/day, based upon increases
in mean absolute and relative liver and kidney weights
PESTICIDE
TOLERANCE:
Leaf petioles subgroup 4B - which include -
All at 3.5 ppm
|
| amaranth,
leafy |
celtuce |
| cardoon
|
fennel,
florence |
| celery
|
rhubarb
|
| celery,
chinese |
swiss
chard |
PESTICIDE
TOLERANCE:
Vegetable, root, except sugar beet, subgroup 1B, except
radish
All
at 0.10 ppm
|
| beet,
garden |
chicory
root |
rutabaga |
| burdock,
edible |
ginseng |
salsify |
| carrot |
horseradish |
salsify,
black |
| carrot
culls |
parsley,
turnip rooted |
salsify,
spanish |
| celeriac
|
parsnip
|
skirret |
| chervil,
turnip rooted |
radish,
oriental
FAN
Note: Under subgroup 1B, "radish" is listed.
If EPA was specific on its exception, then "radish,
oriental" would not be exempt. Needs clarification. |
turnip |
|
| March
5, 2003 |
OPP-2003-0047 |
BAYER,
IR-4. Petition
for Pesticide Tolerance for residues
in or on the raw agricultural commodities vegetable,
root, except sugar beet, subgroup 1B, except radish at 0.1 ppm
and leafy petiole subgroup 4B at 2.0 ppm.
-- EPA has determined that trifloxystrobin
parent and its metabolite CGA-321113 are the residues of concern
for tolerance setting purposes.
-- The liver appears to be the major primary
target organ based on the chronic studies
conducted in mice, rats, and dogs...
-- Genotoxicity. The only positive finding was seen in
the in vitro test system (chinese hamster V79 cells) as a
slight increase in mutant frequency at
a very narrow range (250 - 278 micrograms/milliliter ([mu]g/ml)
of cytologic and precipitating concentrations...
-- In the rat teratology study, reductions
in body weight gain and food consumption were observed
in the dam at >=100 mg/kg... increased incidence of enlarged
thymus, which is a type of variation, at 1,000 mg/kg.
The developmental no observed adverse effect level (NOAEL) was
100 mg/kg.
-- In the rabbit teratology study, body
weight loss and dramatically reduced food consumption
were observed in the dam at >=250 mg/kg. No teratogenic effects
or any other effects were seen on pregnancy or fetal parameters
except for the increase in skeletal anomaly
of fused sternebrae-3 and -4 at the top dose level of
500 mg/kg...
-- In the 2-generation rat reproduction study, body
weight gain and food consumption were
decreased at >=750 ppm, especially
in females during lactation. Consequently, the reduced
pup weight gain during lactation (>=750 ppm) and the
slight delay in eye opening (1,500
ppm) are judged to be a secondary effect of maternal toxicity.
No other fetal effects or any reproductive changes were noted.
The low developmental NOAEL, 50 ppm (5 mg/kg), seen in this
study was probably due to the lack of intermediate dose levels
between 50 and 750 ppm. Based on an evaluation of the dose-response
relationship for pup weight at 750 ppm and 1,500 ppm,
the NOAEL should have been nearly ten-fold higher if such a
dose was available. Based on all these teratology and reproduction
studies, the lowest NOAEL for developmental toxicity is 5 mg/kg
while the lowest NOAEL in the subchronic and chronic studies
is 2.5 mg/kg/day (from the rat chronic study).
... In subchronic studies, several mortality related changes
were reported for the top dose in dogs (500 mg/kg) and rats
(800 mg/kg). At these dose levels, excessive
toxicity has resulted in body weight loss and mortality
with the associated and nonspecific changes in several organs
(such as atrophy in the thymus, pancreas,
bone marrow, lymph node, and spleen) which are not considered
specific target organs for the test compound.
-- In the dog, specific effects were limited to hepatocellular
hypertrophy at >=150 mg/kg and hyperplasia
of the epithelium of the gall bladder at 500 mg/kg. Target
organ effects in the rat were noted as hepatocellular hypertrophy
(>=200 mg/kg) and the related liver weight
increase (>=50 mg/kg). In the mouse,
target organ effects included single cell necrosis (>=300
mg/kg) and hypertrophy (1,050 mg/
kg) in the liver and extramedullary hematopoiesis
(>=300 mg/kg) and hemosiderosis
in the spleen (1,050 mg/kg).
|
| May
22, 2002 |
OPP-2002-
0052 |
BAYER
- Pesticide
Tolerance. FINAL RULE. This regulation establishes
tolerances for trifloxystrobin regulated as trifloxystrobin
and the free form of its acid metabolite CGA-321113 in or
on fruit, stone, group (2 ppm); nut, tree, group (o.04 ppm);
pistachio (0.04 ppm); corn, field, grains (0.05 ppm); corn,
field, forage (0.2 ppm); corn field stover (7 ppm); corn,
field, refined oil (0.1 ppm); corn, pop, grain (0.05 ppm);
corn, pop, stove (7 ppm)r; rice, grain (3.5 ppm); rice, hulls
(8 ppm); rice, straw (7.5 ppm); citrus, dried pulp (0.8 ppm);
citrus oil (30 ppm); fruit, citrus, group (0.3 ppm) ; egg
(0.04 ppm); poultry, fat (0.04 ppm); poultry, meat (0.04 ppm);
and poultry, meat by products (0.04 ppm). |
| Nov
14, 2001 |
PF-1048 |
BAYER
- Pesticide
Petition to Establish Tolerances in or on: barley grain
at 0.05 ppm, straw at 0.05 ppm, barley hay at 0.2 ppm; citrus
fruits crop group at 0.3 ppm, citrus oil at 7.0 ppm; corn
grain at 0.05 ppm, corn forage at 0.05 ppm, corn stover at
7.0 ppm; aspirated grain fractions at 0.1 ppm, popcorn grain
at 0.05 ppm, popcorn stover at 7.0 ppm; rice grain at 3.5
ppm, rice straw at 7.5 ppm; tree nuts crop group at 0.05 ppm;
stone fruits crop group at 2.0 ppm; poultry (fat, kidney,
liver, meat by-products, meat) at 0.05 ppm; and pistachio
at 0.05 ppm. Docket control number PF-1048 |
| August
31, 2001 |
PF-1040 |
BAYER
- Petition
for Pesticide Residue Tolerances in or on the raw agricultural
commodity barley grain at 0.05 ppm, straw at 0.05 ppm, barley
hay at 0.2 ppm, citrus fruits group at 0.3 ppm, citrus oil
at 7.0 ppm, corn forage at 0.05 ppm, corn stover at 7.0 ppm,
aspirated grain fractions at 0.1 ppm, popcorn grain at 0.05
ppm, popcorn stover at 7.0 ppm, pecans at 0.05 ppm, rice grain
at 3.5 ppm, rice straw at 7.5 ppm, stone fruits crops group
at 2.0 ppm, and poultry (fat, kidney, liver, meat by-products,
meats at 0.5 ppm). |
| Dec
20, 2000 |
OPP-181078 |
Pesticide
Emergency Exemptions. In Idaho, US EPA authorized use on
hops to control powdery mildew; 4/13/00 to 9/22/00. In Oregon,
US EPA authorized use on hops to control powdery mildew; 4/13/00
to 9/22/00. In Washington, US EPA authorized use on hops to
control powdery mildew; 4/13/00 to 9/22/00. |
| May
19, 2000 |
PF-912A |
NOVARTIS
- Petition
for Tolerances for the combined residues of trifloxystrobin
and its metabolite, CGA-321113, in or on the raw agricultural
commodities almond nutmeat at 0.04 parts per million (ppm);
almond hulls at 3.0 ppm; hops dried cones at 11.0 ppm; sugar
beet roots at 0.1 ppm; sugar beet tops at 4.0 ppm; sugar beet
dried pulp at 0.4 ppm; sugar beet molasses at 0.2 ppm; potato
tubers at 0.04 ppm; fruiting vegetables at 0.5 ppm; wheat
grain at 0.05 ppm; wheat forage at 0.3 ppm; wheat hay at 0.2;
wheat straw at 5.0 ppm; wheat bran 0.15 ppm; and asperated
grain fractions at 5.0 ppm. |
| Jan
19, 2000 |
PF-912 |
NOVARTIS
- Petition
for tolerance for residues in or on the raw agricultural commodities
almond nutmeats at 0.02 ppm, almond hulls at 1.5 ppm, fruiting
vegetables crop group at 0.7 ppm, hops - dried cones at 11
ppm, potato tubers at 0.02 ppm, sugarbeet roots at 0.05 ppm,
sugarbeet tops at 2.5 ppm, sugarbeet dried pulp at 0.25 ppm,
wheat grain at 0.05 ppm, wheat forage at 0.03 ppm, wheat hay
at 0.2 ppm, wheat straw at 0.05 ppm, and wheat aspirated grain
fractions at 0.5 ppm. |
| Sept
27, 1999 |
OPP-300922 |
NOVARTIS
- Pesticide
Tolerance. - FINAL RULE. This regulation establishes
tolerances for trifloxystrobin regulated as trifloxystrobin
and the free form of its acid metabolite CGA-321113 in or
on pome fruit, cucurbit vegetables, grapes, raisins, peanuts,
peanut hay, wet apple pomace, milk, meat, fat and meat by-
products of cattle, goats, hogs, horses and sheep and bananas.
|
| August
26, 1999 |
PF-819A |
NOVARTIS
- Amended
Petition to increase the tolerance levels for residues in
or on the raw agricultural commodities pome fruit at 0.50
ppm, cucurbit vegetables at 0.50 ppm, grapes at 2.0 ppm, raisins
at 5.0 ppm, peanuts at 0.05 ppm, peanut hay at 4.0 ppm, wet
apple pomace at 5.0 ppm, milk at 0.02 ppm, meat, fat and meat
by-products of cattle, goats, hogs, horses and sheep at 0.05
ppm and imported bananas at 0.1 ppm. |
| April
8, 1999 |
OPP-30471 |
- NOVARTIS
- Application
to Register 3 pesticide products.
- File
Symbol: 100-ORO. Product Name: CGA-279202 WG. Fungicide.
Active ingredient: Trifloxystrobin 50.0%. Proposed classification/Use:
General. For control of certain diseases on pome fruits,
peanuts, grapes, curcurbits, and bananas.
- File
Symbol: 100-ORI. Product Name: CGA-279202 Technical. Fungicide.
Active ingredient: Trifloxystrobin 98%. Proposed classification/Use:
General. For formulating use only.
- File
Symbol: 100-OEN. Product Name: CGA-279202 WG Turf. Fungicide.
Active ingredient: Trifloxystrobin 50.0%. Proposed classification/Use:
General. For use on turf grass.
|
| August
17, 1998 |
PF-819 |
NOVARTIS
- Pesticide
Tolerance Petition for residues of CGA-279202 in or on
the raw agricultural commodity on pome fruit at 0.4 ppm, cucurbit
vegetables at 0.25 ppm, grapes at 1.5 ppm, peanuts at 0.02
ppm, peanut hay at 4.0 ppm, apple pomace at 1.5 ppm and imported
bananas at 0.1 ppm. |
|