|
Return to Potassium
Bifluoride Index Page
ACTIVITY: Wood
Preservative (Inorganic)
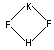
Structure:
Adverse
Effects:
Bone
Chemical Weapons Precursor for the production
of sarin-family nerve agents
Kidney
Lung
Mutagenic
Uses
• for manufacturing wood preservatives
• in the production of soldering agents and brazing.
• as a catalyst for polymerization.
• as a component of electrolyte and fluorine production
• in glass industry for etching and manufacturing
special optical glasses.
|
Potential
Health Effects :
There is limited information available on the hazards of
this chemical. It is assumed that
it will behave similarly to other soluble fluoride salts.
If inhaled or swallowed, this compound can cause fluoride
poisoning. Early symptoms include nausea, vomiting, diarrhea,
and weakness. Later effects include central
nervous system effects, cardiovascular effects and death.
Inhalation:
May cause irritation and burns to
the respiratory tract, symptoms may include coughing,
sore throat, and labored breathing. May be absorbed through
inhalation of dust; symptoms may parallel those from ingestion
exposure. Irritation and burning effects may not appear
immediately.
Ingestion:
May cause salivation, nausea, vomiting, diarrhea, and abdominal
pain, followed by weakness, tremors, shallow respiration,
convulsions, and coma. May cause brain
and kidney damage. Death may be caused by respiratory paralysis.
Affects heart and circulatory system.
Chronic
Exposure:
Chronic exposure may cause mottling
of teeth and bone damage (osteosclerosis) and fluorosis.
Symptoms of fluorisis include brittle bones,weight loss,
anemia, calcified ligaments, general ill health and joint
stiffness.
Aggravation
of Pre-existing Conditions:
Populations that appear to be at increased risk from the
effects of fluoride are individuals that suffer from diabetes
insipidus or some forms of renal impairment.
Ref:
POTASSIUM BIFLUORIDE. Material Data Safety Sheet.
http://www.kingwaychem.com/MSDSflu7.html
|
Bone
(click on for all fluorinated
pesticides)
Ingestion: May cause osseous fluorosis
(increased radiographic density of the bones). May cause
kidney damage, asthma and symptoms resembling
rheumatism. Target Organ Effects:
Chronic ingestion may cause kidney damage.
Ref: Material Safety Data Sheet for Potassium
Hydrogendifluoride [synonym]. LA-CO INDUSTRIES, Inc./Markal Co.
Product Name: SILVER BRAZING FLUX PASTE Revision #: 1.5 Date Prepared:
March 1, 1995. Date Revised: August 6, 2002.
http://www.fluorideaction.org/pesticides/potassium.bifluoride.msds.pdf
Chronic exposure
may cause mottling of teeth and bone damage
(osteosclerosis) and fluorosis. Symptoms of fluorisis include
brittle bones,weight loss, anemia, calcified ligaments, general
ill health and joint stiffness.
Ref:
POTASSIUM BIFLUORIDE. Material Data Safety Sheet.
http://www.kingwaychem.com/MSDSflu7.html
Chemical
Weapons Precursor for the production of sarin-family nerve agents
(click
on for all fluorinated pesticides)
Bifluorides:
Ammonium bifluoride, Potassium bifluoride, Sodium bifluoride.
Bifluorides
are used as a source of the fluorine atom in the synthesis of
all of the G-type nerve agents except Tabun,
in which the fluorine atom is replaced by a cyanide group. All
bifluorides are synthesized from ammonium bifluoride. Ammonium
bifluoride is in turn made from ammonium fluoride which is made
by the reaction of ammonium hydroxide with hydrofluoric acid (HF.)
Ammonia is manufactured on an extremely large scale (>10 million
tons per annum in the US) using the Haber process, for which Fritz
Haber (who played a major role in the German chemical weapons
program in World War I) won a Nobel Prize. Worldwide hydrogen
fluoride manufacture is approximately 400,000 tons. The quantities
needed for manufacture of a stockpile of G agent would be miniscule
in comparison.
Ammonium fluoride is converted to the bifluoride by dehydrating
an aqueous solution of ammonium fluoride. The other bifluorides
are manufactured by essentially the same process, except that
the water, and the more volatile ammonia, are driven off in the
presence of a sodium or potassium compound.
Ref: Nerve
Agent Precursors: Bifluorides: Ammonium bifluoride, Potassium
bifluoride, Sodium bifluoride.
.. some of the precursor chemicals which are early in the production
process and/or are widely produced in industry (and hence not
considered suitable for effective monitoring under the CWC [Chemical
Weapons Convention]) have been included on the AGL [Australia
Group List], because they are either known or suspected to have
been sought for CW purposes. Such precursors include: ...the fluoride
chemicals ... for the production of sarin-family nerve agents...
14 [potassium fluoride]
24 [hydrogen fluoride]
41 [potassium bifluoride]
42 [ammonium bifluoride]
43 [sodium bifluoride]
44 [sodium fluoride]
Ref: A COMPARISON OF THE AUSTRALIA GROUP
LIST OF CHEMICAL WEAPON PRECURSORS AND THE CWC SCHEDULES OF CHEMICALS
by Robert J. Mathews. September 1993. Quarterly Journal of the
Harvard Sussex Program on CBW Armament and Arms Limitation. Issue
No. 21.
http://www.fluoridealert.org/pesticides/Chemical.Weapon.Precursors.pdf
1995
UN Monitoring and Verification of Iraq's Compliance. The organofluorines
on this list include (pesticides
highlighted in red):
List
A (Precursors):
Hydrogen fluoride
(7664-39-3)
Potassium fluoride (7789-23-3)
Ammonium bifluoride (1341-49-7)
Sodium bifluoride (1333-83-1)
Sodium fluoride (7681-49-4)
Potassium bifluoride (7789-29-9)
Fluorine (7782-41-4)
List B:
Sarin (107-44-8)
Soman
(96-64-0)
DF (676-99-3)
PFIB
(382-21-8).
Also included are fluoropolymers
(e.g. Aflex COP, Aflon COP 88, F 40, Ftorlon,
Ftoroplast, Neoflon, ETFE, Teflon, PVDF, Tefzel, PTFE, PE TFE
500 LZ, Haller).
Ref:
1995
- Chemical
& Biological Weapons. Fluorine chemicals.
Kidney
(click on for all fluorinated
pesticides)
Ingestion: May cause
osseous fluorosis (increased radiographic
density of the bones). May cause
kidney damage, asthma and symptoms resembling rheumatism.
Target Organ Effects: Chronic ingestion
may cause kidney damage.
Ref: Material Safety Data Sheet for Potassium
Hydrogendifluoride [synonym]. LA-CO INDUSTRIES, Inc./Markal Co.
Product Name: SILVER BRAZING FLUX PASTE Revision #: 1.5 Date Prepared:
March 1, 1995. Date Revised: August 6, 2002.
http://www.fluorideaction.org/pesticides/potassium.bifluoride.msds.pdf
Lung
(click
on for all fluorinated pesticides)
Ingestion: May cause osseous fluorosis
(increased radiographic density of the bones). May cause
kidney damage, asthma and symptoms
resembling rheumatism. Target Organ Effects: Chronic ingestion
may cause kidney damage.
Ref: Material Safety Data Sheet for Potassium
Hydrogendifluoride [synonym]. LA-CO INDUSTRIES, Inc./Markal Co.
Product Name: SILVER BRAZING FLUX PASTE Revision #: 1.5 Date Prepared:
March 1, 1995. Date Revised: August 6, 2002.
http://www.fluorideaction.org/pesticides/potassium.bifluoride.msds.pdf
Mutagenic
(click on for all fluorinated
pesticides)
Abstract: The L5178Y
mouse lymphoma cell forward-mutation assay was used to test for
the mutagenic activity of sodium and potassium fluoride at the
thymidine kinase locus. Mutants were detected by colony formation
in soft agar in the presence of trifluorothymidine.
Mutagenic and toxic responses were observed in the concentration
range of 300-600 micrograms/ml with both sodium and potassium
fluoride. Approximately 3-fold increases in mutant frequency
were observed for concentrations in the 500-700 micrograms/ml
range that reduced the relative total growth to approximately
10% in the absence or presence of a rat-liver S9 activation system.
A sample of 30% sodium fluoride-70% sodium
bifluoride (NaHF2) induced a similar mutagenic response
but was more toxic with respect to the fluoride concentration.
A specificity for fluoride ions in causing mutagenesis was indicated
by the fact that much higher concentrations of sodium or potassium
chloride were necessary to cause toxicity and increases in the
mutant frequency. The possible involvement of chromosomal changes
was signaled by the predominant increase in the small colony class
of mutants.
Ref: Caspary WJ et al. (1987).
Mutagenic
activity of fluorides in mouse lymphoma cells. Mutat Res.
Mar;187(3):165-80.
|

