|
Return to Lactofen
Adverse Effects
ACTIVITY:
Herbicide (Diphenyl
ether)
CAS Name:
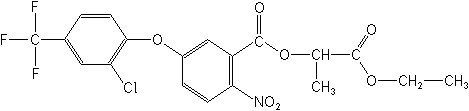
2-ethoxy-1-methyl-2-oxoethyl
5-[2-chloro-4-(trifluoromethyl)phenoxy]-2-nitrobenzoate
Structure:
| Adverse
Effects:
Blood
Body Weight Decrease
Bone
Cancer:
Probable Human Carcinogen - LIVER
Endocrine: Adrenal
Endocrine:
Ovary
Endocrine:
Testicular
Endocrine: Thyroid
Endocrine:
Thymus
Eye
Kidney
Liver
Reproduction
Spleen |
| Environmental
Effects:
Phototoxic
Fish:
Moderately to highly toxic |
Regulatory
Information
(only comprehensive for the US) |
| US
EPA Registered: |
Yes |
| US
EPA PC Code: |
128888 |
| California
Chemical Code |
3538 |
| US
Tolerances: |
CFR
180.432
Note
that CFR 180.453 was
also given to Lactofen. However, as of February 2005, there
is no CFR
180.453 listed
at the Code of Federal Regulations website.
|
| FDA
LMS Code: |
A38 |
US
EPA Permit Date
and Registrant: |
1987,
PPG |
Registered
use in
(includes only a limited list of countries)
|
India,
US, Vietnam
|
US
Maximum Residue Levels permitted
in food commodities
|
Residue
tolerances for Lactofen were amended in Sept 2004.
Two
of Lactofen's metabolites containing the diphenyl ether linkage
were eliminated in the new tolerances. The
established tolerances for snap bean and soybean were reduced
to 0.01 ppm (from 0.05 ppm) as required by the Lactofen Tolerance
Reassessment.
Beans,
snap, succulent (excluding limas)
Cotton, gin byproducts
Cotton, undelinted seed
Peanut
Soybean, seed |
| Other
Information |
| Molecular
Formula: |
C19H15Cl
F3 NO7 |
| Entry
Year: |
1980 |
| Inventing
Company: |
PPG |
| Manufacturers: |
Cyanamid
/ Valent, Aventis |
| Other
Names: |
Cobra
PPG 844
Stellar |
| Of
special interest: |
| PAN
BAD ACTOR - Carcinogen |
| US
Map of Pesticide Use - 1992-1995 |
| 2001
- Tolerance
Reassessment and Reregistration |
| March
12, 2001 -
Report of the Mechanism
of Toxicity Assessment Review Committee -
US EPA |
| March
7, 2001 -
Phototoxic Pesticides:
US EPA Memo Requesting Phototoxicity Study
Protocol for Light-Dependent Peroxidizing Herbicides |
| October
12, 2000 -
Preliminary Human Health
Risk Assessment for Tolerance Reassessment incorporating Revised
Cancer Unit Risks - US EPA |
| July
12, 2000 - Revised
Lactofen (Cobra) Quantitative Risk Assessment based on CD-1
Mouse Dietary Study - US EPA |
May
24, 2000 -
Cancer Assessment
Document. Evaluation of the Carcinogenic
Potential of Diclofop-Methyl. (Second Review). Final Report.
Cancer Assessment Review Committee, Health Effects Division,
US EPA Office of Pesticide Programs. "There are eight
diphenyl ethers that are structurally similar to diclofop-methyl.
Of the chemicals, fomesafen sodium,
haloxyfop-methyl (Verdict), oxyfluorfen,
acifluorfen sodium, nitrofen,
and lactofen were
reviewed in the initial CPRC report. All of these chemicals
induced liver adenomas and carcinomas in rats and/or mice..."
-- organofluorine pesticides highlighted
in red |
| May
4, 2000 - Tolerance
Reassessment of Lactofen: Product and Residue Chemistry Considerations
- US EPA |
| March
2, 2000 -
Toxicology Evaluation - US EPA |
| 2000
Toxic Release Inventory (TRI): brief
summary. |
| Lactofen:
(Cobra Herbicide, PPG-844) Extoxnet Profile |
| Classified
by the State of California: "Known to Cause Cancer" |
| April
2000 - Food
and Drug Administration Pesticide Residue Monitoring. -
Table 3. Pesticides detectable by methods used in 1999 regulatory
monitoring. |
| October
2001 - Glossary
of Pesticide Chemials. A listing of
pesticides subject to analysis of residues in foods and feeds
by the US Food and Drug Administration. |
Material
Safety Data Sheet for Stellar
(R) Herbicide.
Active ingredients: Flumiclorac
Pentyl (7.6%) and Lactofen (26.6%).
-- Flumiclorac is an organofluorine |
Rationale
for US EPA to add Lactofen to the Toxic Release Inventory
Lactofen
(5-(2-chloro-4-(trifluoromethyl)phenoxy)-2-nitro-2- ethoxy-1-methyl-2-oxoethyl
ester) (CAS No. 077501-63-4) (FIFRA AI) (Ref. 3). Lactofen
meets the criteria of an EPA Group B2 compound, i.e., a
probable human carcinogen. This conclusion was based on
an increased incidence of hepatocellular carcinomas in males
and combined incidence of hepatocellular adenomas and carcinomas
in both sexes of CD-1 mice following dietary administration
of lactofen. In CD rats, there was increased incidence of
liver neoplastic nodules in both sexes. Four structurally
similar chemicals, acifluorfen, nitrofen, oxyfluorfen, and
fomesafen, all produced hepatocellular tumors in rodents.
Results
of several subchronic and chronic studies indicated the
liver and kidney as target organs for lactofen. Increased
absolute and relative liver weight and hepatocytomegaly
(the LOEL was 1.5 mg/kg/day; the NOEL was not determined)
were observed in male mice fed lactofen for 78 weeks. At
37.5 mg/kg/day, there was also an increased incidence of
cataracts and renal pigmentation. Based on the LOEL, an
oral RfD of 0.002 mg/kg/day was derived. Renal dysfunction
and decreased hemoglobin and hematocrit levels and red blood
cell counts (the LOEL was 25/75 mg/ kg/day; the NOEL was
5 mg/kg/day) were observed in a 1-year feeding study in
dogs. Increased renal and hepatic pigmentation (the LOEL
was 50 mg/kg/day; the NOEL was 25 mg/kg/day) were noted
in a 2-year feeding study in rats. In a 90-day mouse study,
increased alkaline phosphatase, serum glutamate oxaloacetate
transaminase (SGOT), and serum gleutanic pyruvic transaminase
(SGPT) activities, increased liver weight, hepatic necrosis,
biliary hyperplasia, decreased hematocrit and hemoglobin
levels and red blood cell counts, extramedullary hematopoiesis,
and kidney nephrosis and fibrosis (the LOEL was 26 mg/kg/day;
the NOEL was not determined) were seen. Decreased hemoglobin
and hematocrit levels, decreased red blood cell counts,
and brown pigment in the kidney and liver (the LOEL was
50 mg/kg/day) were noted in a 90-day feeding study in rats.
EPA
believes that there is sufficient evidence for listing lactofen
on EPCRA section 313 pursuant to EPCRA section 313(d)(2)(B)
based on the available carcinogenicity data and hepatic,
renal, and hematological toxicity data for this chemical.
Ref:
USEPA/OPP. Support Document for the Addition of Chemicals
from Federal Insecticide, Fungicide, Rodenticide Act (FIFRA)
Active Ingredients to EPCRA Section 313. U. S. Environmental
Protection Agency, Washington, DC (1993).
As cited by US EPA in: Federal
Register: January 12, 1994. Part IV. 40 CFR Part 372.
Addition of Certain Chemicals; Toxic Chemical Release Reporting;
Community Right-to-Know; Proposed Rule.
|
Based on mechanistic studies with transgenic mice, lactofen
has been classified as a non-genotoxic hepatocarcinogen
in rodents with peroxisome proliferation being a plausible
mode of action. Lactofen is currently classified
as likely to be carcinogenic to humans at high enough doses
to cause the biochemical and histopathological changes in
the liver of rodents, but unlikely to be carcinogenic to
humans below those doses causing these changes.
Ref: Sept 24,
2004, US EPA. Final
Rule for Pesticide Tolerances.
Note:
"Fate data for lactofen show that it has a high binding
potential and that it rapidly is transformed to acifluorfen.
It is not clear whether bound lactofen can be degraded and
released as aciflorfen..."
Ref: June
8, 2001, US EPA, Reregistration
of sodium acifluorifen, p 3
Lactofen is a member of
the diphenyl ether group of herbicides, which includes acifluorfen
(lactofen's major metabolite), nitrofen, oxyfluorfen, and
fomefasen. In addition, lactofen degrades to acifluorfen
in the environment. The Agency has evidence that these compounds
induce similar toxic effects but has not yet determined
whether these compounds exhibit a common mechanism of toxicity.
The Agency
[US EPA] defers the cumulative risk assessment of lactofen
and the other diphenyl ethers to a later date.
Ref: Sept 24, 2004, US EPA. Final
Rule for Pesticide Tolerances.
|
US
Federal Register
••
Note: Due to length, the following is a partial
list. Click here
to see full list of FR entries.
|
| Published
Date |
Docket
Identification Number |
Details |
| June 20, 2007
|
EPA-HQ-OPP-2006-0178
|
IR-4.
Pesticide
Tolerance. FINAL RULE.
| Okra |
0.02 ppm |
Vegetables, fruiting, group
08
This group includes
17 commodities.
chili, postharvest • eggplant • groundcherry
• pepino • pepper • pepper, bell
• pepper, nonbell • pepper, nonbell, sweet
• tomatillo • tomato • tomato, concentrated
products • tomato, dried pomace • tomato,
paste • tomato, puree • tomato, wet pomace
• vegetable, fruiting • vegetable, fruiting,
group
|
0.02 ppm |
DOCUMENTS MADE AVAILABLE WITH
THIS FINAL RULE:
• Lactofen:
Company Notice of Filing. Docket ID: EPA-HQ-OPP-2006-0178-0002
• Lactofen
Acute, Chronic, and Cancer Aggregate Dietary and Drinking
Water Exposure and Risk Assessments for the Section 3 Registration
Action. Docket ID: EPA-HQ-OPP-2006-0178-0004
• Drinking
water and aquatic exposure water assessments for IR4 Tolerance
petition for the new use (R17) of lactofen on the fruiting
vegetable group and okra. Docket ID: EPA-HQ-OPP-2006-0178-0005
• Lactofen.
Addition of New Uses: Fruiting Vegetables (Crop Group 8)
and Okra. PRIA R17. Summary of Analytical Chemistry and
Residue Data. Docket ID: EPA-HQ-OPP-2006-0178-0006
• Data
Evaluation Record. Docket ID: EPA-HQ-OPP-2006-0178-0007
• Lactofen:
Revised Human Health Risk Assessment for Proposed Uses on
Fruiting Vegetables and Okra. Docket ID: EPA-HQ-OPP-2006-0178-0008
• Cancer. Lactofen
has been classified as ``not likely'' to be carcinogenic in
humans because of available data on lactofen support activation
of the peroxisome proliferator activated receptor alpha (PPARa)
as the mode of action which induced liver tumors in rodents.
While the proposed mode of action for
liver tumors in rodent is qualitatively possible in humans,
it is quantitatively implausible and unlikely to take place
in humans based on quantitative species toxicodynamic differences
in PPARa activation. The quantification
of risk is not required.
• Exposure assessment for acifluorfen.
Lactofen degrades in the environment to acifluorfen. Sodium
acifluorfen is a registered agricultural pesticide. Accordingly,
an aggregate assessment for acifluorfen exposure resulting
from both use of lactofen and sodium acifluorfen was also
conducted.
• EPA determined that an additional
safety factor was needed to address the lack of a NOAEL in
the rabbit developmental study. Although sufficient reliable
information has been submitted on developmental effects of
lactofen in rabbits, no NOAEL was identified in one of the
two rabbit developmental studies submitted.
The endpoints of concern identified in available studies are:
Decreased live young/ litter, increased embryonic death/litter,
and increased incidence of post-implantation loss. These effects
were noted at all dose levels (5, 15, 50 mg/kg/day) thus a
NOAEL was not established. Consequently, a LOAEL to
NOAEL factor is appropriate and the risk assessment applies
a 3X uncertainty factor. A FQPA uncertainty factor of infants
and children and will be used for the LOAEL to NOAEL extrapolation.
The 3X factor is considered to be protective because the incidence
of the effects at the lowest dose tested was only marginally
higher than the historical controls.
For sodium acifluorfen, the available
toxicology database provides sufficient information for selecting
various toxicity endpoints and doses for assessing the risks.
The Agency evaluated the hazard and exposure data for sodium
acifluorfen and recommended retaining
the safety factor at 10X due to the data gap for the developmental
neurotoxicity study in rats. In accordance with the current
EPA policy, the 10x factor will be applied to all exposure
durations.
• The drinking water assessment of lactofen
is complicated by the fact that lactofen has a major degradate
in common with another registered herbicide, sodium acifluorfen.
Lactofen and sodium acifluorfen also have common use sites.
The Agency considered the contribution of acifluorfen as an
environmental degradate of lactofen and from sodium acifluorfen
in the aggregate assessment. The drinking water residues used
in the dietary risk assessment were incorporated directly
into this dietary exposure from drinking water assessment.
Therefore, EPA estimated drinking water concentrations for
both lactofen and acifluorfen from lactofen applications. |
| April 13, 2007 |
EPA-HQ-OPP-2007-0005 |
Notice
of Receipt of Requests to Voluntarily Cancel Certain Pesticide
Registrations.
| Registration No. |
Product Name |
Registrant |
| 059639 WA-01-0006 |
Cobra Herbicide |
Valent U.S.A.
Corp.
PO Box 8025
Walnut Creek, CA 94596 |
|
| February 2, 2007 |
EPA-HQ-OPP-2005-0287 |
Pesticide
Registration Review; New Dockets Opened for Review and Comment.
EPA is opening the public comment period for lactofen's registration
review. Registration review is EPA's periodic review of pesticide
registrations to ensure that each pesticide continues to satisfy
the statutory standard for registration, that is, the pesticide
can perform its intended function without unreasonable adverse
effects on human health or the environment.
| Document Date |
Document |
| Jan 2007 |
Summary
Document.
-- Preliminary Work Plam
-- Fact Sheet
-- Ecological Risk Assessment Problem Formulation
-- Human Health Effects Scoping Document
-- Glossary of Terms and Abbreviations
Docket Number: EPA-HQ-OPP-2005-0287-0002
(45 pages) |
| Dec 19, 2006 |
Human
Health Assessments. Scoping document to support registration
review
Docket Number: EPA-HQ-OPP-2005-0287-0003
(9 pages) |
| Dec 13, 2006 |
Problem
Formulation for lactofen registration review
-- the methods that will likely be used in the ecological
risk assessment of lactofen
-- anticipated LOC exceedances
-- data gaps
-- additional data needs
Docket Number:
EPA-HQ-OPP-2005-0287-0004 (26
pages) |
| Oct 18, 2006 |
Screening-level
usage analysis (SLUA)- Available estimates used on
US agricultural crops
Docket Number: EPA-HQ-OPP-2005-0287-0005
(2 pages) |
| Oct 31, 2005 |
Appendix
A - Food/Feed & Non-Food/Non–Feed Uses Eligible
for Registration - Partial Listing.
Docket Number: EPA-HQ-OPP-2005-0287-0006
(2 pages) |
| Oct 25, 2005 |
Section
3, IR4 Tolerance petition for the new use of lactofen
as Cobra Herbicide 2EC on the fruiting vegetable group
and okra.
Docket Number: EPA-HQ-OPP-2005-0287-0007
(9 pages) |
| Jan 8, 2007 |
Human
Health Risk Assessment for Proposed Uses on Fruiting
Vegetables and Okra
Docket Number: EPA-HQ-OPP-2005-0287-0008
(39 pages) |
| Sept 2003 |
Lactofen
TRED Fact Sheet
Docket Number: EPA-HQ-OPP-2005-0287-0009
(4 pages) |
| Sept 24, 2003 |
Report
of the Food Quality Protection Act (FQPA) Tolerance Reassessment
Progress and Risk Management Decision (TRED) for Lactofen
Docket Number: EPA-HQ-OPP-2005-0287-0010
(9 pages) |
| Oct 12, 2000 |
Preliminary
Human Health Risk Assessment for Tolerance Reassessment
Incorporating Revised Cancer Unit Risks
Docket Number: EPA-HQ-OPP-2005-0287-0011
(30 pages) |
| May 4, 2000 |
Tolerance
Reassessment of Lactofen: Product and Residue Chemistry
Considerations
Docket Number: EPA-HQ-OPP-2005-0287-0012
(38 pages) |
| Jan 21, 2003 |
Drinking
Water Exposure Assessment for Lactofen, Updated for
Prospective Ground Water (PGW) Monitoring Study
Docket Number: EPA-HQ-OPP-2005-0287-0013
(40 pages) |
| July 14, 2000 |
Revised
Drinking Water Exposure Assessment for Lactofen
Docket Number: EPA-HQ-OPP-2005-0287-0014
(34 pages) |
| July 22, 2004 |
Occupational
and Residential Risk Assessment for Lactofen on Cotton
and Peanuts
Docket Number: EPA-HQ-OPP-2005-0287-0015
(27 pages) |
|
| April 12, 2006 |
EPA-HQ-OPP-2006-0178 |
IR-4.
Pesticide
petition; New Tolerance PP 5E6930.
in or on various food commodities
-- vegetable, fruiting, group at 0.01 pp
This group (group 8) includes
17 commodities.
chili, postharvest • eggplant • groundcherry
• pepino • pepper • pepper, bell •
pepper, nonbell • pepper, nonbell, sweet • tomatillo
• tomato • tomato, concentrated products •
tomato, dried pomace • tomato, paste • tomato,
puree • tomato, wet pomace • vegetable, fruiting
• vegetable, fruiting, group
-- okra at 0.01 ppm. |
| Sept
24, 2004 |
OPP-2004-0293 |
Valent.
Pesticide Tolerance.
FINAL RULE.
The proposed and established tolerances are corrected to conform
to the Food and Feed Commodity Vocabulary Database and
to lower the established tolerances for snap
bean and soybean to 0.01 ppm as required by the Lactofen Tolerance
Reassessment. Section
180.432 is revised to read as follows:
Sec. 180.432 Lactofen; tolerances for residues.
(a) Tolerances are established for residues of the herbicide
lactofen, 1-(carboethoxy)ethyl 5-[2-chloro-4-(trifluoromethyl)phenoxy]-
2- nitrobenzoate, in or on the following raw agricultural
commodities:
| Commodity
|
Parts per
million |
| Beans, snap, succulent
(excluding limas) |
0.01 |
| Cotton, gin byproducts |
0.02 |
| Cotton, undelinted seed |
0.01 |
| Peanut |
0.01 |
| Soybean, seed |
0.01 |
| Table 2.--Summary
of Toxicological Dose and Endpoints for lactofen for Use
in Human Risk Assessment |
| Exposure Scenario |
Dose Used in Risk
Assessment, Interspecies and Intraspecies and any Traditional
UF |
Special FQPA SF and Level of Concern
for Risk Assessment
|
Study and Toxicological Effects |
| Acute Dietary (Females 13-50 years of
age) |
NOAEL = 50 mg/kg/day UF
= 100 Acute RfD = 0.5 mg/kg/day |
Special FQPA SF = 3 aPAD = acute RfD/
Special FQPA SF = 0.17 mg/kg/day |
Rat Developmental Toxicity Study
LOAEL = 150 mg/kg/day based on decreased
fetal weight and skeletal abnormalities. |
| Acute Dietary (General population including
infants and children). |
An endpoint attributable
to a single dose (exposure) was not identified from the
available studies, including the developmental toxicity
studies in rats and rabbits. |
| Chronic Dietary (All populations) |
NOAEL = 0.79 mg/kg/day
UF = 100 Chronic RfD = 0.008 mg/kg/day |
Special FQPA SF = 1 cPAD = chronic RfD/
Special FQPA SF = 0.008 mg/kg/day |
Dog chronic toxicity LOAEL = 3.96 mg/kg/day
based on increased incidence of
proteinaceous casts in the kidneys,
and
statistically significant increases
in the absolute weights of the thyroid and adrenal glands
in males. |
| Cancer (Oral, dermal, inhalation) |
Lactofen
acts via a peroxisome proliferation mechanism of action.
Likely to be carcinogenic to humans at high enough doses
to cause these biochemical and histopathological effects
(peroxisome proliferation) in the livers of rodents but
unlikely to be carcinogenic at doses below those causing
these changes. Lactofen
is considered to be a threshold carcinogen. NOAEL
= 0.3 mg/kg/day based on increased
activities of liver enzymes and increased incidence of
liver histopathological findings at the LOAEL of 1.5 mg/kg/day. |
••
The toxicology database for lactofen
is complete for FQPA purposes except for a developmental toxicity
study in rabbits. Based on the quality
of the exposure data, EPA determined
that the 10X SF to protect infants and children should be
reduced to 3X
•• The available rabbit
developmental toxicity study was considered unacceptable because
dosing was not done at a high enough level to observe significant
toxicity.
••
Endpoints for other risk assessments (chronic and cancer)
utilize NOAELs significantly lower than 20mg/kg/day; therefore
the developmental rabbit study will not affect these assessments.
Based on
mechanistic studies with transgenic mice, lactofen has been
classified
as a non-genotoxic hepatocarcinogen in rodents with peroxisome
proliferation being a plausible mode of action. Lactofen
is currently
classified as likely to be carcinogenic to humans at high
enough doses
to cause the biochemical and histopathological changes in
the liver of
rodents, but unlikely to be carcinogenic to humans below those
doses
causing these changes. |
| Jan
28, 2004 |
OPP-2003-0294 |
Report
of the Food Quality Protection Act (FQPA) Tolerance Reassessment
and Risk Management Decision (TRED)
for Lactofen; Notice of Availability.
• This document announces availability of and starts
a 30-day public comment period for the FQPA TRED for Lactofen.
•
Tolerances for lactofen in or on raw
agricultural commodities for plants are currently established
for the combined residues of lactofen and its associated metabolites
containing the diphenyl ether linkage, but will be revised
to include only lactofen per se. The
two existing tolerances for lactofen have been reassessed
and will be lowered from 0.05 ppm to 0.01 ppm. There
are currently no tolerances for lactofen in processed commodities
or animal commodities, and the available residue data indicate
that tolerances for these commodities are not necessary.
Documents
available:
1.
Sept 2003: Lactofen
TRED Facts - (4 pages)
2.
Sept 24, 2003: Report
of the Food Quality Protection Act (FQPA) Tolerance Reassessment
Progress and Risk Management Decision (TRED) for Lactofen
- (9 pages)
3.
Sept 24, 2003: Overview
of Lactofen FQPA Risk Assessment for Tolerance Reassessment
- (9 pages)
4.
Aug 12, 2003: Lactofen.
Revisions to HED Tolerance Reassessment Risk Assessment
- (3 pages)
5.
May 22, 2002: Lactofen
- Report of the Cancer Assessment Review Committee - (38
pages)
6.
March 12, 2001: LACTOFEN:
Report of the Mechanism of Toxicity Assessment Review Committee
- (17 pages)
7.
Feb 26, 2003: EFED
Review of Lactofen Small-Scale Prospective Ground-Water Monitoring
Study 166-1 DER MRID #456717-01, 02, and -03. - (40
pages)
8.
Jan 21, 2003: DRINKING
WATER EXPOSURE ASSESSMENT FOR LACTOFEN, UPDATED FOR
PROSPECTIVE GROUND WATER (PGW) MONITORING STUDY - (40
pages)
9.
Sept 15, 2003: Addendum
to EFED RED Chapter for sodium acifluorfen. Addendum to TRED
for lactofen - (41 pages) |
••
Note: Due to length, the above is a partial list.
Click here
to see full list of FR entries.
|
|