|
Return to
Adverse Effects
Abstracts
ACTIVITY: Herbicide
(pyridine)
CAS Name:
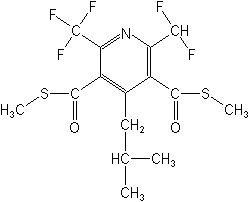
S,S?-dimethyl
2-(difluoromethyl)-4-(2-methylpropyl)-6-(trifluoromethyl)-3,5-pyridinedicarbothioate
Structure:
| Adverse
Effects:
Body
Weight Decrease
Endocrine: Adrenal
Endocrine: Ovary
Endocrine: Testicular
Endocrine: Thymus
Endocrine: Thyroid
Endocrine: Uterus
Kidney
Liver
Lung
Spleen
|
| Environmental
Effects:
Fish:
Moderately to Highly Toxic
Zooplankton:
Highly Toxic |
Regulatory
Information
(only comprehensive for the US) |
| US
EPA Registered: |
Yes |
| US
EPA PC Code: |
128994 |
| California
Chemical Code |
2308 |
US
EPA Permit Date
and Registrant: |
1991,
Monsanto |
Registered
use in
(includes only a limited list of countries)
|
Australia,
Canada, US |
| Other
Information |
| Molecular
Formula: |
C15H16
F5 NO2S2 |
| Entry
Year: |
1987 |
| Inventing
Company: |
Monsanto |
| Manufacturers: |
Monsanto
Rohm & Haas |
| Other
Names: |
Dimension,
Stakeout,
MON-7200,
MON-15100 |
| Manufacture
site: |
US:
Rohm & Haas, Tyrone,
Pennsylvania 16686 |
| Of
special interest: |
| PAN
Data |
| Material
Safety Data Sheets & Labels |
2005
- New Jersey: "On April 22, parents of Toms River High
School East students received a letter notifying them that the
athletic fields were going to be sprayed with pesticides today,
during spring break. The letter stated that the fields will
be treated with Dithiopyr, dimethylamine
and glyphosate and this warning follows: "harmful if swallowed,
inhaled or absorbed through the skin, avoid contact with eyes
or clothing."
It goes on to warn that the Office of Pesticide Programs of
the Environmental Protection Agency has stated: "Where
possible, persons who potentially are sensitive, such as pregnant
women, infants and children, should avoid any unnecessary exposure."
...
Letter
in the Asbury Park Press 4/27/05. |
| February
1993 - Summary
of Toxicology Studies With Dithiopyr. Dennis
P. WARD, Toxicology Department, The Agricultural Grop, A Unit
of Monsanto Company. |
| Rohm
and Haas - Canadian Material
Safety Data Sheet for Dimension herbicide (1999). |
| Abstracts
|
| 1988
- 1992 -
US EPA Office of Cleared
Reviews, 1988 to 1992. |
| June
2002 - In Australia when Dithiopyr is used for "Treatment
of turf where grazing by, or feeding to, livestock including
poultry will not occur" no
maximum residue levels are required. Ref: June 2002. Table 5.
Uses of substances where maximum residue limits are not necessary.
Australian National Registration Authority for Agricultural
Veterinary Chemicals. The MRL Standard. Maximum residue limits
in food and animal feedstuff.
http://www.nra.gov.au/residues/mrl5.pdf
|
Rationale
for US EPA to add Dithiopyr to the Toxic Release Inventory
In
a 2-generation rat reproduction study, decreased body weight,
diffuse hepatocellular swelling, and ``white spots'' on
the livers were observed in the offspring of rats administered
greater than or equal to 16.4 mg/kg/day. The NOEL values
were 1.7 mg/kg/day. In a 13-week rat feeding study, the
LOEL of 6.62 mg/kg/day produced diffuse hepatocellular swelling.
The NOEL was 0.662 mg/kg/day. In a 13-week dog feeding study,
increased alkaline phosphatase, discolored livers, and cholestasis
was observed at 10 mg/kg/day (LOEL). The NOEL was 1 mg/kg/day.
In addition, at 30 mg/kg/day, increased serum glutamic-pyruvic
transaminase and serum glutamic oxaloacetic transaminase,
increased liver and kidney weights, and decreased cholesterol
and albumin were observed. EPA believes that there is sufficient
evidence for listing dithiopyr on EPCRA section 313 pursuant
to EPCRA section 313(d)(2)(B) based on the available hepatic
and renal toxicity data.
Ref: USEPA/OPP. Support Document for the
Addition of Chemicals from Federal Insecticide, Fungicide,
Rodenticide Act (FIFRA) Active Ingredients to EPCRA Section
313. U. S. Environmental Protection Agency, Washington,
DC (1993).
As cited by US EPA in: Federal
Register: January 12, 1994. Part
IV. 40 CFR Part 372. Addition of Certain Chemicals; Toxic
Chemical Release Reporting; Community Right-to-Know; Proposed
Rule.
Note:
Addition
of Certain Chemicals; Toxic Chemical Release Reporting;
Community Right-to-Know. Final
Rule.
Dithiopyr
was included in the deferral of 40 chemicals: EPA
is deferring final action on 40 chemicals and one chemical
category until a later date. These chemicals and the comments
received on them raised particularly difficult technical
or policy issues which will require additional time to address.
The Agency does not believe that it would be in the spirit
of community right-to-know to delay final action on the
remaining 286 chemicals and chemical categories, pending
completion of work on the more limited group. In a future
rulemaking, EPA will make a final determination as to whether
these chemicals should be added to EPCRA section 313. The
public comment that has been received specific to these
deferred chemicals will be addressed as part of the future
rulemaking discussed above.
|
| US
Federal Register |
| Date
Published |
Docket
Identification Number |
Details |
| August
17, 2005 |
OPP-2005-0222 |
Requests
to Voluntarily Cancel Certain Pesticide Registrations.
|
Registration no. |
Product
Name |
Chemical
Name |
Registrants
Requesting Voluntary Cancellation |
| 010404-00097 |
Superstar
Plus Mini Fertilizer |
Dithiopyr |
Lesco
Inc., 1301 E. 9th Street,
Suite 1300, Clevela, OH
441141849. |
|
| December
13, 1999 |
OPP-300939 |
Clomazone;
Pesticide Tolerances for Emergency Exemptions. Final Rule.
... Clomazone is a member of the pyridazones/pyridinones
class of herbicides. Other members of this class include purazon,
norflurazon, fluridone, oxadiazon, fluorochloridone, amitrol,
and dithiopyr. EPA does not have,
at this time, available data to determine whether clomazone
has a common mechanism of toxicity with other substances or
how to include this pesticide in a cumulative risk assessment... |
| November
6, 1996 |
40 CFR
Part 455
[FRL-5630-9]
RIN 2040-AC21 |
Pesticide
Chemicals Category, Formulating, Packaging and Repackaging
Effluent Limitations Guidelines, Pretreatment Standards, and
New Source Performance Standards.
Final Rule.
| Table
10 to Part 455.--List of Appropriate Pollution Control
Technologies \1\ |
| PAI
name \2\ |
Shaughnessy
code \4\ |
Structural group \5\ |
Treatment
technology |
| Dithiopyr
(ANSI) |
128994 |
Pyridine |
Activated
Carbon |
\1\ The
272 Pesticide Active Ingredients (PAIs) are listed first,
by PAI code, followed by the non-272 PAIs from the 1988 FIFRA
and TSCA Enforcement System (FATES) Database, which are listed
in Shaughnessy code order. PAIs that were exempted or reserved
from the PFPR effluent guidelines are not listed in the table.
\2\ The
non-272 PAI names are taken directly from the 1988 FATES database.
Several of the PAI [pesticide active ingredient] names are
truncated because the PAI names listed in the FATES database
are limited to 60 characters.
\4\ All
Shaughnessy codes are taken from the 1988 FATES database.
Some of the 272 PAIs are not listed in the 1988 FATES database;
therefore, no Shaughnessy codes are listed for these PAIs.
\5\ Structural
groups are based on an analysis of the chemical structures
of each PAI. |
| November
30, 1994 |
OPPTS-400082B7 |
Addition
of Certain Chemicals; Toxic Chemical Release Reporting; Community
Right-to-Know. Final
Rule.
EPA is
deferring final action on 40 chemicals and one chemical category
until a later date. These chemicals and the comments received
on them raised particularly difficult technical or policy
issues which will require additional time to address. The
Agency does not believe that it would be in the spirit of
community right-to-know to delay final action on the remaining
286 chemicals and chemical categories, pending completion
of work on the more limited group. In a future rulemaking,
EPA will make a final determination as to whether these chemicals
should be added to EPCRA section 313. The public comment that
has been received specific to these deferred chemicals will
be addressed as part of the future rulemaking discussed above.
These chemicals follow:
dithiopyr |
| Jan 12,
1994 |
OPPTS-400082 |
EPA's
proposal to add 41
fluorine and organofluorine chemicals to the Toxics Release
Inventory (TRI). See excerpt in box
above. Also available at http://www.epa.gov/tri/frnotices/59fr1788.htm |
|