|
Return to
Adverse Effects
Abstracts
ACTIVITY:
Herbicide (anilide)
CAS Name:
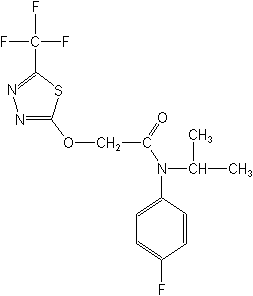
N-(4-fluorophenyl)-N-(1-methylethyl)-2-[[5-(trifluoromethyl)-1,3,4-thiadiazol-2-yl]oxy]acetamide
Structure:
| Adverse
Effects:
Blood
Body
Weight Decrease
Bone
Brain
Cholesterol
Endocrine: Thyroid
Endocrine: Uterus
Eye
Heart
Kidney
Liver
Sciatic
Nerve
Spinal Cord
Spleen
|
| Environmental
Effects:
Highly
toxic to terrestrial, semi-aquatic, and aquatic plants.
Likely to persist in soil for some time |
Regulatory
Information
(only comprehensive for the US) |
| US
EPA Registered: |
Yes |
| US
EPA PC Code: |
121903 |
| California
Chemical Code |
5293 |
| US
Tolerances: |
CFR
180.527 |
| FDA
LMS Code: |
B30 |
Registered
use in
(includes only a limited list of countries)
|
Canada,
Germany, Hungary, Philippines, South Africa, US, UK |
US
Maximum Residue Levels permitted
in food commodities
|
Permitted in or on over
500 food commodities,
including: Alfalfa, Cattle, Clover, Corn, Goat, Hog, Horse,
Sheep, Soybean, Wheat |
UK
Temporary MRL's
|
Applicable
from: June 2001
Potato:
0.1 p pm
Wheat & Barley (winter): 0.05 ppm |
| Other
Information |
| Molecular
Formula: |
C14H13
F4 N3O2S |
| Entry
Year: |
1998 |
| Manufacturers: |
Bayer |
| Other
Names: |
Artist,
Axiom DF, Domain, Epic, Fluthiamide, Herold, Terano,
FOE 5043 Technical,
FOE 5043 DF, Thiafluamide |
| Of
special interest: |
| PAN
Data |
| Material
Safety Data Sheets & Labels |
| April
26, 2005 - U.S. Congressman Emmanuel Cleaver (D-MO) introduced
a bill "to extend the temporary suspension of duty
on Flufenacet (FOE Hydroxy) " from Dec. 31, 2006 to Dec.
31, 2008. The bill was referred to the House Committee on Ways
and Means. |
| July
2003 -
Review report for the active substance flufenacet. Flufenacet
7469/VI/98-Final. European Commission.
Finalised in the Standing Committee on the Food Chain and Animal
Health at its meeting on 4 July 2003 in view of the inclusion
of flufenacet in Annex I of Directive 91/414/EEC. |
| April
8, 1998 -
Pesticide Fact Sheet.
- US EPA |
April
2002 -
Beer in the UK: 23 organofluorine
pesticides approved for use on malting barley.
Published by the British Beer and Pubs
Association and Brewing Research International. |
| Abstracts |
February
18, 2002 -
European Commission
decision to extend provisional authorisation granted
for new active substances including flufenacet |
| March
15, 2000 - BAYER -
Press Release on
new $45 million facility at Kansas City headquarters to manufacture
flufenacet, the active ingredient in Axiom, Axiom¨ AT, Domainª
and Epicª DF Herbicides. |
| August
2001 -
IR-4: New
Products/Transitional Solution List - This
list contains brief descriptions of numerous new pest control
materials that have been introduced over the last several years.
Additionally, it contains information on some "older"
crop protection chemicals that are believed to have room for
new uses. This List includes: Flufenacet
|
Metabolites
/ Breakdown products of Flufenacet
Ref: Pesticide Action Network |
| PAN
Data |
1,3,4-Thiadiazol-2(3H)-one,
5-(trifluoromethyl) |
CAS No.
84352-75-0 |
| PAN
Data |
3-Glucosyl-5-trifluoromethyl-1,3,4-thiadiazol-2(3H)-one
|
No CAS
No. |
| Note
from FAN: US
EPA tolerances for residues of flufenacet on food commodities
include metabolites containing the 4-fluoro-N-methylethyl
benzenamine moiety. |
"Based
on the toxicological findings for flufenacet relating to
endocrine disruption effects, flufenacet should be considered
as a candidate for evaluation as an endocrine disrupter
when the criteria are established."
Ref.
US EPA:Federal
Register. March 29, 2000.
|
US
Federal Register
••
Note: Due to length, the following is a partial
list. Click here
to see full list of FR entries.
|
| Published
Date |
Docket
Identification Number |
Details |
| May 9, 2007 |
EPA-HQ-OPP-2006-0965 |
Bayer CropScience.
Pesticide
Tolerance. FINAL RULE.
EPA determined that additional tolerances
are needed in connection with the petitioned-for tolerances
for wheat bran 0.80 ppm, grass forage at 7.0 ppm, and grass
hay at 0.4 ppm, cattle kidney at 0.05 ppm, goat kidney
at 0.05 ppm, hog kidney at 0.05 ppm, horse kidney at 0.05
ppm, and sheep kidney at 0.05 ppm. EPA determined that tolerance
levels are needed that differ from those proposed by the registrant
for sweet corn forage at 0.45 ppm (0.4 ppm proposed) sweet
corn stover at 0.30 ppm, (0.4 ppm proposed) wheat forage at
6.0 ppm (10.0 ppm proposed), wheat grain at 0.60 ppm (1.0
ppm proposed), wheat hay at 1.2 ppm (2.0 ppm proposed), and
wheat straw at 0.35 ppm (0.5 ppm proposed). EPA determined
that tolerances are not necessary for fat, meat, and meat
byproducts of cattle, goat, hog, horse, and sheep. Since permanent
tolerances are being established for wheat
and kidney of cattle, goat, hog, horse, and sheep, emergency
exemption tolerances for these commodities are being deleted.
Prenatal and postnatal sensitivity.
According to EPA: There is no indication of additional susceptibility
of young rats or rabbits following prenatal exposure to flufenacet
in the developmental toxicity studies. There was an indication
of qualitative susceptibility in the 2-generation reproduction
study. Effects seen in the offspring in the reproductive
toxicity studies (including increased
pup death in early lactation and cannibalism) were more severe
than those seen in the parental animals (increased liver weight
and cytomegaly), although there was no difference in
the NOAELs/LOAELS between parental animals and offspring in
that study. Increased susceptibility
(qualitative and quantitative) was seen in the developmental
neurotoxicity study in rats. Decreased
body weight was seen in pups at all dose levels, and additional
effects, including decreased motor activity, delayed developmental
landmarks, and decreases in morphometric measurements were
seen at mid and high doses. Morphometric measurements
were not made at the low dose. A slight decrease in body weight
in mid and high dose dams during early lactation may have
been due to palatability of test substance and was not considered
adverse.
Conclusion. Several factors
weighed in favor of the conclusion that no additional safety
factor is needed to protect the safety of infants and children.
First, there was no evidence of increased susceptibility in
the developmental toxicity studies (rats and rabbits), and
qualitative susceptibility seen in the
rat reproduction study did not raise concerns because the
pup death may be attributable to maternal cannibalism,
and there was a clear NOAEL for the effect. Second, there
are also no additional residual uncertainties with respect
to exposure data:
| General Tolerances
are established for the combined residues of the herbicide
flufenacet, N-(4-fluorophenyl)-N-(1-methylethyl)-2-[[5-(trifluoromethyl)-1,
3, 4-thiadiazol-2-yl] oxy]acetamide and its metabolites
containing the 4-fluoro-N-methylethyl benzenamine moiety
in or on the following commodities. |
| Commodity |
Parts per million |
| Cattle, kidney |
0.05 |
| Corn, field, forage |
0.4 |
| Corn, field, grain. |
0.05 |
| Corn, field, stover |
0.4 |
| Corn, sweet, forage |
0.45 |
| Corn, sweet, kernel plus cob with husks
removed |
0.05 |
| Corn, sweet, stover |
0.30 |
| Goat, kidney |
0.05 |
| Hog, kidney |
0.05 |
| Horse, kidney |
0.05 |
| Sheep, kidney |
0.05 |
| Soybean, seed |
0.1 |
| Wheat, bran |
0.80 |
| Wheat, forage |
6.0 |
| Wheat, grain |
0.60 |
| Wheat, hay |
1.2 |
| Wheat, straw |
0.35 |
Tolerances with regional
registrations. Tolerances are established for
combined residues of flufenacet, N-(4-fluorophenyl)-N-(1-methylethyl)-2-[[5-(trifluoromethyl)-1,
3, 4-thiadiazol-2-yl]
oxy]acetamide, and its metabolites containing the 4-fluoro-N-
methylethyl benzenamine moiety, with regional registration. |
| Commodity |
Parts per million |
| Grass, forage |
7.0 |
| Grass, hay |
0.4 |
|
••
Note: Due to length, the above is a partial list.
Click here
to see full list of FR entries.
|
|