|
Return
to Picolinafen Index Page
Activity:
Herbicide
(Pyridinecarboxylic
acid)
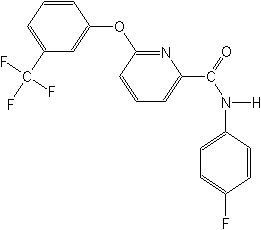
Structure:
Adverse Effects:
Anemia
Blood
Body Weight Decrease
Bone
Endocrine:
Adrenal gland
Endocrine: Thyroid
Liver
Spleen
Environmental
Anemia
and
Blood
(click
on for all fluorinated pesticides)
-- In the rat 2-generation reproduction study, reproduction function,
reproductive parameters and litter parameters were not influenced
by treatment in the first and second generation (P1/P2) parental
animals at any dose level up to and including 500 ppm (equal to
39 and 42 mg/kg bw/d in males and females, respectively), the
highest dose tested. Hematological findings,
increased spleen weights, and histopathological findings indicative
of regenerative hemolytic anemia
were noted for P1/P2 males and females at 250 ppm (equal to 19
and 21 mg/kg bw/d for males and females, respectively) and above.
Hematological findings including lower red
blood cell count, hemoglobin, and hematocrit were also
noted for male and female second generation (F2) pups at 250 ppm
and above on lactation day 21 (only time point evaluated). Although
the hematological findings noted in the F2 offspring may be secondary
to maternal toxicity, a direct treatment-related effect cannot
be dismissed; therefore, these findings
were considered to be toxicologically relevant. The NOAEL
for parental and offspring toxicity was 50 ppm (equal to 3.7 and
4.0 mg/kg bw/d in males and females, respectively). On the basis
of the parental and offspring NOAELs in the rat 2-generation reproductive
toxicity study (one litter/generation), there was no indication
that neonates were quantitatively more sensitive than adults to
the toxic effects of picolinafen.
-- Hematological and histopathological findings indicative of
regenerative hemolytic anemia were noted
in all species tested. The most sensitive species appears
to be the rat. The most appropriate NOAEL for regenerative hemolytic
anemia is 50 ppm (equal to 2.4 and 3.0 mg/kg bw/d for males and
females, respectively), as determined in the 2-year rat dietary
study. The MOE for regenerative hemolytic anemia is 171 compared
to the ADI.
Ref: February 17, 2003 - Canada. Picolinafen.
Regulatory Note REG2003-02. Alternative Strategies and Regulatory
Affairs Division, Pest Management Regulatory Agency.
http://www.fluorideaction.org/pesticides/picolinafen.canada.feb.2003.pdf
Body
Weight Decrease
(click
on for all fluorinated pesticides)
-- In dogs fed picolinafen
at dietary concentrations of 0, 50, 150 or 1500 ppm for 12 months
body weight gains were decreased in all
groups of treated males... Enlarged
thyroids and increased thyroid weights were observed at 1500 ppm.
Increased follicular hypertrophy in the thyroid was observed in
both sexes at 1500 ppm and hyperplasia in the thyroid was observed
in females at 1500 ppm. A NOEL could
not be established in this study since body weight gain was reduced
at all tested doses. The LOEL in this study was 50 ppm,
equal to 1.4 mg/kg bw/day in males and 1.6 mg/kg bw/day in females.
Ref: Public Release Summary on Evaluation
of the new active PICOLINAFEN in the products SNIPER HERBICIDE
& PARAGON HERBICIDE. Australia. National Registration Authority
for Agricultural and Veterinary Chemicals November 2000.
http://www.nra.gov.au/publications/prspic.pdf
Determination of Acceptable Daily Intake. The most appropriate
NOAEL of 1.4 mg/kg bw/d in the 1-year dietary study in dogs is
recommended as the basis for the acceptable daily intake (ADI).
Treatment-related findings at the lowest observed adverse effect
level (LOAEL), the next highest dose level, included lower
body weight and body-weight gain for males (approximately 20 and
48%, respectively). A safety factor of 100 to account for
intraspecies and interspecies variations was considered to be
adequate; no additional safety factors are required. The recommended
ADI is 0.014 mg/kg bw/d.
Ref:
Nov 2005 - AC 900001 (Picolinafen). Proposed Regulatory Decision
Document. PRDD2005-5. Canadian
Pest Management Regulatory Agency (PMRA).
http://www.fluorideaction.org/pesticides/picolinafen.canada.nov05.pdf
Bone
(click
on for all fluorinated pesticides)
-- In a developmental
study, picolinafen was given to rabbits by oral gavage at 0, 5,
20 or 50 mg/kg bw/day on days 6-28 of gestation... The number
of resorptions was slightly increased at 50 mg/kg bw/day and an
increased incidence of fused sternal centra
was observed in foetuses at 50 mg/kg bw/day. The NOEL for maternal
toxicity in this study was 5 mg/kg bw/day and the NOEL for embryotoxicity
and foetotoxicity was 20 mg/kg bw/day.
-- -- Picolinafen was given to pregnant rats by oral gavage at
0, 100, 500 or 1000 mg/kg bw/day on days 6 to 19 of gestation...
The incidence of bipartite ossification
in the thoracic vertebrae in foetuses at 1000 mg/kg bw/day
was increased, but no other alterations were observed. Since a
NOEL for maternal toxicity was not established in this study,
a follow-up study using 0, 5, 25 or 50 mg/kg bw/day was conducted.
No treatment-related effects were observed on dams or on foetal
development... In a developmental study, picolinafen was given
to rabbits by oral gavage at 0, 5, 20 or 50 mg/kg bw/day on days
6-28 of gestation. Soft or liquid faeces were observed at 50 mg/kg
bw/day and body weight gain and food consumption were reduced
at 20 and 50 mg/kg bw/day... The number of resorptions was slightly
increased at 50 mg/kg bw/day and an increased incidence of
fused sternal centra was observed in foetuses at 50 mg/kg
bw/day. The NOEL for maternal toxicity in this study was 5 mg/kg
bw/day and the NOEL for embryotoxicity and foetotoxicity was 20
mg/kg bw/day.
Ref: Public Release Summary on Evaluation
of the new active PICOLINAFEN in the products SNIPER HERBICIDE
& PARAGON HERBICIDE. Australia. National Registration Authority
for Agricultural and Veterinary Chemicals November 2000.
http://www.nra.gov.au/publications/prspic.pdf
Histopathological findings were generally characterized by increased
incidence/severity of hemosiderin deposition in the spleen and/or
Kupffer cells of the liver and extramedullary hematopoiesis in
the spleen and/or liver. Under normal physiological conditions
a certain amount of hemosiderin deposition is routinely observed
in the spleen due to normal breakdown of effete red blood cells;
however, for all species tested, increased incidence/severity
were noted for both sexes at the higher dose levels. The increased
incidence/severity of extramedullary hematopoiesis was most likely
a compensatory response to the increased hemolysis of red blood
cells noted for all species tested. Other histopathological findings
associated with regenerative hemolytic anemia included
increased erythropoietic activity in the bone marrow and
liver at higher doses. This was noted in rats following 28-day
dietary administration at 1000 ppm and was indicated by a change
in the myeloid:erythroid ratio from 2:1 in the controls to 1:1
in rats at 1000 ppm. For males, this shift to a more immature,
stronger population of red cells at 1000 ppm was considered to
account for the slight but significant decrease in erythrocyte
osmotic fragility. For females, this correlated with an increased
incidence of erythropoiesis in the bone
marrow (femur/joint and sternum).
Ref: February 17, 2003 - Canada.
Picolinafen. Regulatory Note REG2003-02. Alternative Strategies
and Regulatory Affairs Division, Pest Management Regulatory Agency.
http://www.fluorideaction.org/pesticides/picolinafen.canada.feb.2003.pdf
Endocrine:
Adrenal
(click
on for all fluorinated pesticides)
In the 78-week dietary study, there was no evidence to indicate
that picolinafen was oncogenic in the mouse. In the rat 2-year
dietary study, there was a non-statistically
significant, increased incidence of benign neoplasms (benign pheochromocytomas)
in the adrenal gland medullary region for males at 500 ppm (highest
dose tested). The incidence was within recent historical
control data for benign medullary neoplasms. In addition, there
was no decrease in the time-to-appearance of the induced tumour
and no dose-response relationship in proliferative changes usually
associated with neoplasms, benign or malignant, in the adrenal
medulla. Published literature indicates that proliferative lesions
of the adrenal medulla in male rats occurs at relatively high
incidences and can be spontaneous (age-related) in nature. Based
on these findings, the slight increased incidence of benign neoplasms
in the adrenal gland medullary region noted for males at 500 ppm,
was most likely spontaneous in nature and not treatment-related.
The weight of evidence suggests that picolinafen is not likely
to be oncogenic in humans.
Ref: February 17, 2003 - Canada. Picolinafen.
Regulatory Note REG2003-02. Alternative Strategies and Regulatory
Affairs Division, Pest Management Regulatory Agency.
http://www.fluorideaction.org/pesticides/picolinafen.canada.feb.2003.pdf
Endocrine:
Thyroid (click
on for all fluorinated pesticides)
-- In a study in which
groups of two dogs per sex were fed picolinafen at dietary concentrations
of 0, 100, 1000, 2000, or 10000 ppm for 28 days, food consumption
and body weight gain were reduced at 10000 ppm...
Thyroid weight was increased in a dose-related manner in
dogs at all doses and thyroid enlargement
accompanied by histopathological changes was observed at ¥ 1000
ppm.
-- In groups of four dogs per sex fed picolinafen at dietary concentrations
of 0, 50, 500 or 2500 ppm for 90 days haemoglobin... Thyroids
were enlarged at 2500 ppm and thyroid weights were increased in
males at ¥500 ppm and in females at 2500 ppm... Follicular hypertrophy
was observed in the thyroid at 500
and 2500 ppm with hyperplasia at 2500 ppm. The NOEL in this study
was 50 ppm equal to 1.7 mg/kg bw/day in males and 1.8 mg/kg bw/day
in females.
-- In dogs fed picolinafen at dietary concentrations of 0, 50,
150 or 1500 ppm for 12 months body weight gains were decreased
in all groups of treated males... Enlarged
thyroids and increased thyroid
weights were observed at 1500 ppm. Increased follicular hypertrophy
in the thyroid was observed in both
sexes at 1500 ppm and hyperplasia in the
thyroid was observed in females at 1500 ppm. A NOEL could
not be established in this study since body weight gain was reduced
at all tested doses. The LOEL in this study was 50 ppm, equal
to 1.4 mg/kg bw/day in males and 1.6 mg/kg bw/day in females.
Ref: Public Release Summary on Evaluation
of the new active PICOLINAFEN in the products SNIPER HERBICIDE
& PARAGON HERBICIDE. Australia. National Registration Authority
for Agricultural and Veterinary Chemicals November 2000.
http://www.nra.gov.au/publications/prspic.pdf
-- Although findings indicative of hemolytic anemia were noted
in dogs following 90-day and 1-year dietary administration, the
main target organ appeared to be the thyroid
as indicated by increased thyroid weight,
diffuse hypertrophy of the thyroid follicular epithelial cells,
and scattered foci of thyroid follicular cell hyperplasia,
at 500 ppm (17.3 and 20.2 mg/kg bw/d and above for males and females,
respectively) and above following 90-day dietary administration
and at 1500 ppm (42.7 and 47.1 mg/kg bw/d for males and females,
respectively) and above following 1-year dietary administration
(the differences noted following 90-day and 1-year dietary administration
were due to dose selection). Hormone levels (thyroxine, tri-iodothyronine
and thyroid stimulating hormone [TSH]) were not determined. Lower
body weight and/or body-weight gain were also noted in dogs at
2500 ppm (equal to 87.5 and 92.1 mg/kg bw/d for males and females,
respectively) following 90-day dietary administration and at 150
ppm (1.7 and 1.8 mg/kg bw/d and above for males and females, respectively)
and above following 1-year dietary administration. The NOAEL following
90-day dietary administration was 50 ppm (equal to 1.7 and 1.8
mg/kg bw/d for males and females, respectively). The NOAEL following
1-year dietary administration was 50 ppm (equal to 1.4 and 1.6
mg/kg bw/d for males and females, respectively).
-- The treatment-related findings noted in the
thyroid (increased thyroid weight, diffuse hypertrophy of the
thyroid follicular epithelial cells, and scattered foci of thyroid
follicular cell hyperplasia) in dogs following 90-day and
1-year dietary administration may be suggestive of a neurotoxicity
potential. Similar lesions were not observed in the rat (including
neonates) or mouse following subchronic or chronic dietary exposure
and there was no other evidence in any species tested to indicate
a neurotoxicity potential. Thyroid hormone (thyroxine, tri-iodothyronine,
and TSH) levels were not determined. Thyroid hormones are crucial
to normal growth and development in the central nervous system
and in the absence of the hormone, brain development can be retarded;
therefore, in the absence of thyroid hormone data and in the absence
of any human data, these lesions cannot
be disregarded and must be considered relevant to humans.
Ref: February 17, 2003 - Canada. Picolinafen.
Regulatory Note REG2003-02. Alternative Strategies and Regulatory
Affairs Division, Pest Management Regulatory Agency.
http://www.fluorideaction.org/pesticides/picolinafen.canada.feb.2003.pdf
Occupational exposure will be predominately via the dermal route,
and of short- to intermediate-term duration. Although
a 4-week repeat dose dermal toxicity study is available, it is
not considered adequate for occupational and bystander risk assessment
since it does not adequately account for the treatment-related
findings noted in the thyroid in dogs following 90-day and 1-year
dietary administration. To account for these findings,
it is recommended that the dog 90 day dietary study be used for
the proposed exposure scenarios. The recommended NOAEL is 1.7
mg/kg bw/d. A safety factor of 100 to account for intraspecies
and interspecies variations is considered to be adequate; no additional
safety factors were used because there was an adequate MOE to
the NOAEL of 4.4 mg/kg bw/d for thyroid effects in the 1-year
dietary dog study.
Ref:
Nov 2005 - AC 900001 (Picolinafen). Proposed Regulatory Decision
Document. PRDD2005-5. Canadian
Pest Management Regulatory Agency (PMRA).
http://www.fluorideaction.org/pesticides/picolinafen.canada.nov05.pdf
Liver
(click
on for all fluorinated pesticides)
-- Fat, liver
and kidneys contained the highest amounts of picolinafen metabolites
labelled on the pyridine ring, whereas the blood,
liver, spleen and lungs contained the highest amounts of
picolinafen metabolites labelled on the aniline ring.
-- Short-Term Studies All mice fed picolinafen at dietary concentrations
of 0, 100, 1000, 2000, 3500 or 7000 ppm for 28 days... Discolouration
of the extremities, discoloured organs (kidney,
liver, lungs, testes and heart) and haematological changes
(increased MCH, MCHC, reticulocyte and Heinz bodies numbers) were
observed at 3500 and 7000 ppm. Spleen and liver
weights were increased at ¥ 2000 ppm and the breakdown products
of blood cells accumulated in the liver
and spleen and the production of blood cells in the spleen were
increased in males at ¥ 1000 ppm and females at ¥ 2000 ppm. The
severity of these histopathological changes increased in a dose-related
manner.
-- In a study in which groups of two dogs per sex were fed picolinafen
at dietary concentrations of 0, 100, 1000, 2000, or 10000 ppm
for 28 days, ... Livers were enlarged,
liver weights and liver glycogen
increased and serum cholesterol was increased in males at ¥ 1000
ppm and females at ¥ 2000 ppm.
-- In mice fed picolinafen at dietary concentrations of 0, 50,
500, 1000 or 2000 ppm for 13 week... Liver
weight and pigment deposition were increased at 2000 ppm with
other histopathological changes observed in the liver
of males at ¥ 500 ppm and females at ¥1000 ppm. The spleens of
some mice were enlarged and spleen weights were increased in females
at ¥1000 ppm...
- In rats fed picolinafen at dietary concentrations of 0, 80,
400 or 800 ppm ... Liver to body
weight ratios were increased in males at 800 ppm and females at
400 and 800 ppm. Pigment deposition was increased in the liver
and spleen at 400 and 800 ppm. The NOEL in this study was 80 ppm,
equal to 6.4 mg/kg bw/day in males and 6.8 mg/kg bw/day in females.
-- Long-Term Studies In mice fed picolinafen at dietary concentrations
of 0, 40, 400 or 800 ppm for 18 months, there was no increase
in the incidence of neoplasms... Increased liver
weights and histopathological changes were observed in females
at 800 ppm with histopathological changes observed in the
livers of males at 400 and 800 ppm.
Ref: Public Release Summary on Evaluation
of the new active PICOLINAFEN in the products SNIPER HERBICIDE
& PARAGON HERBICIDE. Australia. National Registration Authority
for Agricultural and Veterinary Chemicals November 2000.
http://www.nra.gov.au/publications/prspic.pdf
Following subchronic
and chronic administration, mice also exhibited treatment-related
findings in the liver, including centrilobular
hepatocellular hypertrophy and hepatocellular vacuolation.
These were noted following 28-day, 90-day and 78-week dietary
administration. The NOAELs for mice were 23.4, 10.2, and 6.9 mg/kg
bw/d following 28-day, 90-day, and 78-week dietary administration,
respectively.
Ref:
February 17, 2003 - Canada. Picolinafen. Regulatory Note REG2003-02.
Alternative Strategies and Regulatory Affairs Division, Pest Management
Regulatory Agency.
http://www.fluorideaction.org/pesticides/picolinafen.canada.feb.2003.pdf
Spleen
(click
on for all fluorinated pesticides)
-- Fat, liver and kidneys
contained the highest amounts of picolinafen metabolites labelled
on the pyridine ring, whereas the blood, liver, spleen
and lungs contained the highest amounts of picolinafen metabolites
labelled on the aniline ring.
-- Short-Term Studies. All mice fed picolinafen at dietary concentrations
of 0, 100, 1000, 2000, 3500 or 7000 ppm for 28 days survived,
but body weight gain was reduced in males at 7000 ppm... Spleen
and liver weights were increased at ¥ 2000 ppm and the breakdown
products of blood cells accumulated in the liver and spleen
and the production of blood cells in the spleen
were increased in males at ¥ 1000 ppm and females at ¥ 2000 ppm.
The severity of these histopathological changes increased in a
dose-related manner.
-- In mice fed picolinafen at dietary concentrations of 0, 50,
500, 1000 or 2000 ppm for 13 weeks, food consumption was decreased
in males at 1000 and 2000 ppm... The spleens
of some mice were enlarged and spleen
weights were increased in females at ¥1000 ppm. Pigment deposition
was increased in the spleen at ¥
500 ppm and extramedullary haematopoiesis was increased in females
at ¥ 500 ppm males at ¥1000 ppm. The NOEL in this study was 50
ppm, equal to 10 mg/kg bw/day in males and 13 mg/kg bw/day in
females.
-- In mice fed picolinafen at dietary concentrations of 0, 50,
500, 1000 or 2000 ppm for 13 weeks, food consumption was decreased
in males at 1000 and 2000 ppm... The spleens
of some mice were enlarged and spleen weights were increased in
females at ¥1000 ppm. Pigment deposition was increased in the
spleen at ¥ 500 ppm and extramedullary
haematopoiesis was increased in females at ¥ 500 ppm males at
¥1000 ppm. The NOEL in this study was 50 ppm, equal to 10 mg/kg
bw/day in males and 13 mg/kg bw/day in females.
-- In rats fed picolinafen at dietary concentrations of 0, 80,
400 or 800 ppm for 13 weeks body weight gain was slightly reduced
in females at 800 ppm... Spleen weights
were increased at 400 and 800 ppm in males and 800 ppm in females.
Liver to body weight ratios were increased in males at 800 ppm
and females at 400 and 800 ppm. Pigment deposition was increased
in the liver and spleen at 400 and
800 ppm. The NOEL in this study was 80 ppm, equal to 6.4 mg/kg
bw/day in males and 6.8 mg/kg bw/day in females.
-- Long-Term Studies In mice fed picolinafen at dietary concentrations
of 0, 40, 400 or 800 ppm for 18 months, there was no increase
in the incidence of neoplasms... Splenic
enlargement was slightly increased in incidence in females at
400 and 800 ppm. Extramedullary haematopoiesis was observed in
the spleens of mice at 800 ppm with
increased pigment deposition observed in males at 800 ppm and
females at 400 and 800 ppm. The NOEL in this study was 40 ppm,
equal to 6.9 mg/kg bw/day for males and 8.2 mg/kg bw/day for females.
-- In rats fed picolinafen at dietary concentrations of 0, 50,
250, or 500 ppm for 24 months there was no increase in the incidence
of neoplasms. Haemoglobin, Hct and RBC were reduced at 3 and 6
months only at 250 and 500 ppm. Spleen
weights were increased at 500 ppm and the amount of brown pigment
observed in the spleen was increased
at 250 and 500 ppm at both 12 and 24 months. The NOEL in this
study was 50 ppm, equal to 2.4 mg/kg bw/day for males and 3.0
mg/kg bw/day for females.
-- Rats were fed picolinafen at dietary concentration of 0, 50,
250 or 500 ppm in a two-generation reproduction study... The spleen
also showed congestion, increased amounts of brown pigment and
extramedullary haematopoiesis at 250 and 500 ppm... The NOEL in
this study is 50 ppm, equal to 3.7 mg/kg bw/day for males and
4.2 mg/kg bw/day for females.
-- Picolinafen was given to pregnant rats by oral gavage at 0,
100, 500 or 1000 mg/kg bw/day on days 6 to 19 of gestation. One
dam at 1000 mg/kg bw/day appeared emaciated and body weight losses
associated with reduced food consumption were observed on days
6-12 of gestation at ¥ 500 mg/kg bw/day. Spleen
weights were increased in dams at ¥100 mg/kg bw/day and
liver to body weight ratios were slightly increased at ¥ 500 mg/kg
bw/day... The incidence and severity of
extramedullary haematopoiesis and haemosiderosis in the spleen
was increased in a dose dependent manner in all treated groups...
-- In a developmental study, picolinafen was given to rabbits
by oral gavage at 0, 5, 20 or 50 mg/kg bw/day on days 6-28 of
gestation... Congestion and haemosiderosis in the spleen
were increased in incidence and severity at 20 and 50 mg/kg bw/day
and increased extramedullary haematopoiesis was observed at 50
mg/kg bw/day.
Ref: Public Release Summary on Evaluation
of the new active PICOLINAFEN in the products SNIPER HERBICIDE
& PARAGON HERBICIDE. Australia. National Registration Authority
for Agricultural and Veterinary Chemicals November 2000.
http://www.nra.gov.au/publications/prspic.pdf
| Environmental
(click
on for all fluorinated pesticides)
- Potential
of major transformation product (CL 153815) to leach and
contaminate groundwater (page 71) :
-- The
major transformation product:
CL 153815 is classified as persistent in soil under anaerobic
conditions (page 22). The transformation product,
CL 153815, meets the criterion for persistence in sediment.
The CL 153815 half-life in sediment (645 days) exceeds the
TSMP Track 1 cut-off criterion (≥ 365 days). - (page
44)
Fate
Process for CL 153815 - Major Transformation Product:
(page 71) |
| Anaerobic
soil |
persistent |
| Aerobic
water layer |
slightly
persistent |
| Aerobic
water / anaerobic sediment |
moderately
persistent. |
| Anaerobic
water / anaerobic sediment |
persistent |
| Anaerobic
water layer |
persistent |
| Anaerobic
sediment layer |
persistent |
Ref:
Nov 2005 - AC 900001 (Picolinafen). Proposed Regulatory
Decision Document. PRDD2005-5. Canadian
Pest Management Regulatory Agency (PMRA).
http://www.fluorideaction.org/pesticides/picolinafen.canada.nov05.pdf
|
|

