|
Return
to Lactofen Index Page
Activity:
Herbicide
(Diphenyl
ether)
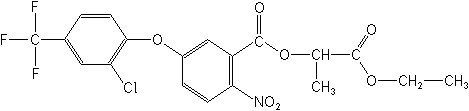
Structure:
Adverse Effects:
Blood
Body Weight Decrease
Bone
Cancer:
Probable Human Carcinogen - LIVER
Endocrine: Adrenal
Endocrine:
Ovary
Endocrine:
Testicular
Endocrine: Thyroid
Endocrine: Thymus
Eye
Kidney
Liver
Reproduction
Spleen
Environmental
As
of February 15, 2005, this herbicide is permitted in
or on 5 food commodities
in the United States. Residue
tolerances for Lactofen were amended in Sept 2004.
Two
of Lactofen's metabolites containing the diphenyl ether
linkage were eliminated in the new tolerances. The
established tolerances for snap bean and soybean were reduced
to 0.01 ppm (from 0.05 ppm) as required by the Lactofen
Tolerance Reassessment. The 5 food
commodities are:
Beans,
snap, succulent (excluding limas) - 0. 01 ppm
Cotton, gin byproducts
- 0. 02 ppm
Cotton, undelinted seed
- 0. 01 ppm
Peanut
- 0. 01 ppm
Soybean, seed
- 0. 01 ppm
-
see tolerances at http://www.fluorideaction.org/pesticides/mrl.lactofen.htm
|
Blood
(click on for all fluorinated
pesticides)
Renal dysfunction and
decreased hemoglobin and hematocrit levels
and red blood cell counts (the LOEL was 25/75 mg/ kg/day;
the NOEL was 5 mg/kg/day) were observed in a 1-year feeding study
in dogs. Increased renal and hepatic pigmentation (the LOEL was
50 mg/kg/day; the NOEL was 25 mg/kg/day) were noted in a 2-year
feeding study in rats. In a 90-day mouse study, increased alkaline
phosphatase, serum glutamate oxaloacetate transaminase (SGOT),
and serum gleutanic pyruvic transaminase (SGPT) activities, increased
liver weight, hepatic necrosis, biliary hyperplasia, decreased
hematocrit and hemoglobin levels and red blood cell counts,
extramedullary hematopoiesis, and kidney nephrosis and fibrosis
(the LOEL was 26 mg/kg/day; the NOEL was not determined) were
seen. Decreased hemoglobin and hematocrit
levels, decreased red blood cell counts, and brown pigment
in the kidney and liver (the LOEL was 50 mg/kg/day) were noted
in a 90-day feeding study in rats. EPA believes that there is
sufficient evidence for listing lactofen on EPCRA section 313
pursuant to EPCRA section 313(d)(2)(B) based on the available
carcinogenicity data and hepatic, renal, and hematological
toxicity data for this chemical.
Ref:
USEPA/OPP. Support Document for the Addition of Chemicals from
Federal Insecticide, Fungicide, Rodenticide Act (FIFRA) Active
Ingredients to EPCRA Section 313. U. S. Environmental Protection
Agency, Washington, DC (1993). As cited by US EPA in: Federal
Register: January 12, 1994. Part IV. 40 CFR Part 372. Addition
of Certain Chemicals; Toxic Chemical Release Reporting; Community
Right-to-Know; Proposed Rule.
Body
Weight Decrease (click
on for all fluorinated pesticides)
Prenatal developmental--
rodents (rat)
Maternal NOAEL = 50 mg/kg/day. Maternal LOAEL = 150 mg/kg/day
based on signs of toxicity (excessive salivation, lethargy, dried
red material around the nares and inguinal regions) and statistically
significant decreases in body weight gain.
Developmental NOAEL = 50 mg/kg/day.
Developmental LOAEL = 150 mg/kg/day based on decreased
fetal weight and skeletal abnormalities (increased incidence
of bent ribs and/or limb bones) and reduced ossification of vertebral
arches.
Ref:
Sept 24, 2004. Lactofen. Pesticide Tolerance. Final Rule. Federal
Register.
http://www.fluorideaction.org/pesticides/lactofen.fr.sept.24.2004.htm
Bone
(click
on for all fluorinated pesticides)
-- Prenatal developmental-- rodents (rat). Developmental NOAEL
= 50 mg/kg/day.
Developmental LOAEL = 150 mg/kg/day based on decreased
fetal weight and skeletal abnormalities
(increased incidence of bent ribs and/or
limb bones) and reduced ossification of vertebral arches.
-- Rat Developmental Toxicity
Study. LOAEL = 150 mg/kg/day based on decreased fetal weight and
skeletal abnormalities.
Ref:
Sept 24, 2004. Lactofen. Pesticide Tolerance. Final Rule. Federal
Register.
http://www.fluorideaction.org/pesticides/lactofen.fr.sept.24.2004.htm
Developmental toxicity--
Rats. Pregnant rats were administered oral doses of 0, 15, 50
and 150 mg/kg/day Lactofen Technical on days 6- 19 of gestation.
Maternal toxicity (death, abortion and reduced body weight gain)
was observed at 150 mg/kg/day. Developmental toxicity (reduced
fetal weight, slightly reduced ossification,
bent ribs and bent limb bones)
was also observed at 150 mg/kg/day. The NOEL for this study was
50 mg/kg/day."
Ref: Federal Register, February 25, 1998.
[PF-789; FRL-5767-5]
http://www.fluoridealert.org/pesticides/Lactofen.FR.Feb.1998.htm
Cancer:
Probable Human Carcinogen - LIVER
(click
on for all fluorinated pesticides)
Likely
to be Carcinogenic to Humans at High Doses. Not
Likely to be Carcinogenic to Humans at Low Doses. Hepatocellular
carcinomas
(M); Hepatocellular adenomas & carcinomas (M & F); CD-mice.
Liver neoplastic nodules; Sprague-Dawley rats (M & F). MOE
approach should be used for estimating human cancer risk, using
a NOAEL of 2 ppm (0.3 mg/kg/day)
Ref: April
26, 2006 . Chemicals Evaluated for Carcinogenic Potential by the
Office of Pesticide Programs. From: Jess Rowland, Chief Science
Information Management Branch Health Effect Division (7509C) Office
of Pesticide Programs, USEPA.
http://www.fluorideaction.org/pesticides/pesticides.cancer.potential.2006.pdf
Group B2--Probable Human Carcinogen.
Reviewed 4/ 8/ 87.
Ref: List of Chemicals Evaluated for Carcinogenic
Potential. Science Information Management Branch, Health Effects
Division, Office of Pesticide Programs, U. S. Environmental Protection
Agency. March 15, 2002.
http://www.biomuncie.org/chemicals_evaluated_for_carcinog.htm
--
Based on mechanistic studies with transgenic mice, lactofen
has been classified as a non-genotoxic hepatocarcinogen in rodents
with peroxisome proliferation being a plausible mode of action.
Lactofen is currently classified as likely to be carcinogenic
to humans at high enough doses to cause the biochemical and histopathological
changes in the liver of rodents, but unlikely to be carcinogenic
to humans below those doses causing these changes.
-- Cancer (Oral, dermal, inhalation).
Lactofen is considered to be a threshold
carcinogen. NOAEL = 0.3 mg/kg/day based
on increased activities of liver enzymes and increased incidence
of liver histopathological findings at the LOAEL of 1.5 mg/kg/day.
Ref: Sept 24, 2004.
Lactofen. Pesticide Tolerance. Final Rule. Federal Register.
http://www.fluorideaction.org/pesticides/lactofen.fr.sept.24.2004.htm
Lactofen meets the
criteria of an EPA Group B2 compound, i.e., a probable human carcinogen.
This conclusion was based on an increased incidence of hepatocellular
carcinomas in males and combined incidence of hepatocellular
adenomas and carcinomas in both sexes of CD-1 mice following
dietary administration of lactofen. In CD rats, there was increased
incidence of liver neoplastic nodules in
both sexes. Four structurally similar chemicals, acifluorfen,
nitrofen, oxyfluorfen, and fomesafen, all produced hepatocellular
tumors in rodents. Results of several subchronic and chronic studies
indicated the liver and kidney as
target organs for lactofen. Increased absolute and relative liver
weight and hepatocytomegaly (the LOEL was 1.5 mg/kg/day;
the NOEL was not determined) were observed in
male mice fed lactofen for 78 weeks. At 37.5 mg/kg/day,
there was also an increased incidence of cataracts and renal pigmentation.
Based on the LOEL, an oral RfD of 0.002 mg/kg/day was derived.
Renal dysfunction and decreased hemoglobin and hematocrit levels
and red blood cell counts (the LOEL was 25/75 mg/ kg/day; the
NOEL was 5 mg/kg/day) were observed in a 1-year feeding study
in dogs. Increased renal and hepatic pigmentation (the LOEL was
50 mg/kg/day; the NOEL was 25 mg/kg/day) were noted in a 2-year
feeding study in rats. In a 90-day mouse study, increased alkaline
phosphatase, serum glutamate oxaloacetate transaminase (SGOT),
and serum gleutanic pyruvic transaminase (SGPT) activities, increased
liver weight, hepatic necrosis, biliary hyperplasia, decreased
hematocrit and hemoglobin levels and red blood cell counts, extramedullary
hematopoiesis, and kidney nephrosis and fibrosis (the LOEL was
26 mg/kg/day; the NOEL was not determined) were seen. Decreased
hemoglobin and hematocrit levels, decreased red blood cell counts,
and brown pigment in the kidney and liver
(the LOEL was 50 mg/kg/day) were noted in a 90-day feeding study
in rats. EPA believes that there is sufficient evidence for listing
lactofen on EPCRA section 313 pursuant to EPCRA section 313(d)(2)(B)
based on the available carcinogenicity data and hepatic,
renal, and hematological toxicity data for this chemical.
Ref:
USEPA/OPP. Support Document for the Addition of Chemicals from
Federal Insecticide, Fungicide, Rodenticide Act (FIFRA) Active
Ingredients to EPCRA Section 313. U. S. Environmental Protection
Agency, Washington, DC (1993). As cited by US EPA in: Federal
Register: January 12, 1994. Part IV. 40 CFR Part 372. Addition
of Certain Chemicals; Toxic Chemical Release Reporting; Community
Right-to-Know; Proposed Rule.
Chronic/carcinogenicity
feeding study-- Mouse-- 24-month. In a dietary 18-month oncogenicity
study in mice at dosages of 10, 50 and 250 ppm Lactofen Technical,
an increase in liver adenomas and
carcinomas, cataracts and liver pigmentation
was observed at 250 ppm. The lowest dose, 10 ppm, was the LOEL
based on increased liver weight and hepatocytomegaly.
-- Chronic/carcinogenicity feeding study-- Rat-- 24-month. In
a 2- year chronic feeding/oncogenicity study of Lactofen Technical
in rats at dosages of 0, 500, 1,000 and 2,000 ppm in the diet,
an increase in liver neoplastic nodules
and foci of cellular alteration was observed in both sexes at
2,000 ppm. The NOEL for systemic toxicity is 500 ppm based on
kidney and liver pigmentation.
-- Carcinogenicity. The Toxicology Branch Peer Review Committee
has determined that lactofen meets the criterion for a B2 (possible
human) carcinogen since it caused an increase in liver
tumors (adenomas and/or carcinomas) in two species. Based
on the mouse oncogenicity study, a human upper-bound potency estimate
(Q1*) was calculated as 0.17 (mg/kg/day).
-- More importantly, evidence summarized above suggest that carcinogenic
effects observed in rodent liver related to long term lactofen
consumption are attributable to peroxisomal
proliferation as opposed to a direct genotoxic effect.
This mechanism of action would more appropriately be regulated
as a threshold effect (similar to RfD comparisons) as opposed
to a non-threshold effect with a quantitative potency factor derived
from low dose extrapolations. This change in the hazard assessment
process for lactofen would have a profound effect on the exposure
and risk assessments for this chemical.
Ref: Federal Register: February 25, 1998
[Page 9532-9540]. Notice of Filing of Pesticide Petition.
http://www.fluoridealert.org/pesticides/Lactofen.FR.Feb.1998.htm
"There are eight
diphenyl ethers that are structurally similar to diclofop-methyl.
Of the chemicals, fomesafen sodium, haloxyfop-methyl
(Verdict), oxyfluorfen, acifluorfen
sodium, nitrofen, and lactofen
were reviewed in the initial CPRC report. All
of these chemicals induced liver adenomas and carcinomas in rats
and/or mice..."
Ref: May 24, 2000 - Cancer Assessment Document.
Evaluation of the Carcinogenic Potential of Diclofop-Methyl. (Second
Review). Final Report. Cancer Assessment Review Committee, Health
Effects Division, US EPA Office of Pesticide Programs.
http://www.fluoridealert.org/pesticides/Cancer.EPA.Assess.May.2000.pdf
•
Note
from FAN:
Except for Nitrofen, all the pesticides cited above are fluorinated.
Endocrine:
Adrenal
(click
on for all fluorinated pesticides)
Dog chronic toxicity LOAEL = 3.96 mg/kg/day based
on increased incidence of
proteinaceous casts in the kidneys,
and statistically significant increases
in the absolute weights of the thyroid and adrenal glands in males.
Ref:
Sept 24, 2004. Lactofen. Pesticide Tolerance. Final Rule. Federal
Register.
http://www.fluorideaction.org/pesticides/lactofen.fr.sept.24.2004.htm
-- Subchronic feeding--
Mice-- 3-month. Groups of Male and female mice were fed diets
containing Lactofen Technical at concentrations of 0, 40, 200,
1,000, 5,000, and 10,000 for 13-weeks. At week 5, the dosage of
the 40 ppm groups was increased to 2,000 ppm. Treatment related
mortality occurred at dosages above 1,000 ppm. The LOEL was 200
ppm based on: increased WBC; decreased hematocrit, hemoglobin
and RBC; increased alkaline phosphatase, SGOT, SGPT, cholesterol
and total serum protein levels; increased weights or enlargement
of the spleen, liver, adrenals, heart
and kidney; histopathological changes of the liver, kidney,
thymus, spleen, ovaries and testes. In general, effects were slight
in the 200 ppm groups, and moderate to severe in the 1,000 ppm
groups.
Ref: Federal Register: February 25,
1998 [Page 9532-9540]. Notice of Filing of Pesticide Petition.
http://www.fluoridealert.org/pesticides/Lactofen.FR.Feb.1998.htm
Endocrine:
Ovary
(click
on for all fluorinated pesticides)
-- Subchronic feeding--
Mice-- 3-month. Groups of Male and female mice were fed diets
containing Lactofen Technical at concentrations of 0, 40, 200,
1,000, 5,000, and 10,000 for 13-weeks. At week 5, the dosage of
the 40 ppm groups was increased to 2,000 ppm. Treatment related
mortality occurred at dosages above 1,000 ppm. The LOEL was 200
ppm based on: increased WBC; decreased hematocrit, hemoglobin
and RBC; increased alkaline phosphatase, SGOT, SGPT, cholesterol
and total serum protein levels; increased weights or enlargement
of the spleen, liver, adrenals, heart and
kidney; histopathological changes of the liver, kidney, thymus,
spleen, ovaries and testes.
In general, effects were slight in the 200 ppm groups, and moderate
to severe in the 1,000 ppm groups.
Ref: Federal Register: February 25,
1998 [Page 9532-9540]. Notice of Filing of Pesticide Petition.
http://www.fluoridealert.org/pesticides/Lactofen.FR.Feb.1998.htm
Endocrine:
Testicular
(click
on for all fluorinated pesticides)
-- Subchronic feeding--
Mice-- 3-month. Groups of Male and female mice were fed diets
containing Lactofen Technical at concentrations of 0, 40, 200,
1,000, 5,000, and 10,000 for 13-weeks. At week 5, the dosage of
the 40 ppm groups was increased to 2,000 ppm. Treatment related
mortality occurred at dosages above 1,000 ppm. The LOEL was 200
ppm based on: increased WBC; decreased hematocrit, hemoglobin
and RBC; increased alkaline phosphatase,
SGOT, SGPT, cholesterol and total serum protein levels; increased
weights or enlargement of the spleen, liver, adrenals, heart and
kidney; histopathological changes of the liver, kidney, thymus,
spleen, ovaries and testes.
In general, effects were slight in the 200 ppm groups, and moderate
to severe in the 1,000 ppm groups.
-- Reproduction-- Rats. Groups of male and female rats were fed
0, 50, 500 or 2,000 ppm of Lactofen Technical continuously in
their diets for 2-generations. Adult systemic toxicity (mortality,
reduced body weight, increased liver and spleen weight, decreased
kidney weight and histological changes in the liver and testes)
was observed at levels of 500 ppm and greater.
Ref: Federal Register: February 25, 1998
[Page 9532-9540]. Notice of Filing of Pesticide Petition.
http://www.fluoridealert.org/pesticides/Lactofen.FR.Feb.1998.htm
90-Day Feeding - mouse:
LEL=200 ppm (increased leucocytes, decreased Hct, Hgb, WBC); increased
alkaline phosphatase, SGOT, SGPT, cholesterol; increased liver
weight and enlarged spleen, liver and heart; liver vacuolation
necrosis of individual hepatocytes, biliary hyperplasia, extramedullary
hematopoiesis, kidney nephrosis and fibrosis; follicular atreria,
abnormal sperm forms); core grade
minimum (PPG Industries, 1983b)
Ref: US EPA IRIS. (Online October 11, 2003).
Available at Toxnet.
Endocrine:
Thymus
(click
on for all fluorinated pesticides)
-- Subchronic feeding--
Mice-- 3-month. Groups of Male and female mice were fed diets
containing Lactofen Technical at concentrations of 0, 40, 200,
1,000, 5,000, and 10,000 for 13-weeks. At week 5, the dosage of
the 40 ppm groups was increased to 2,000 ppm. Treatment related
mortality occurred at dosages above 1,000 ppm. The LOEL was 200
ppm based on: increased WBC; decreased hematocrit, hemoglobin
and RBC; increased alkaline phosphatase, SGOT, SGPT, cholesterol
and total serum protein levels; increased weights or enlargement
of the spleen, liver, adrenals, heart
and kidney; histopathological changes of the liver, kidney, thymus,
spleen, ovaries
and testes. In general, effects were slight in the 200
ppm groups, and moderate to severe in the 1,000 ppm groups.
Ref: Federal Register: February 25, 1998
[Page 9532-9540]. Notice of Filing of Pesticide Petition.
http://www.fluoridealert.org/pesticides/Lactofen.FR.Feb.1998.htm
Endocrine:
Thyroid (click
on for all fluorinated pesticides)
Dog chronic toxicity LOAEL = 3.96 mg/kg/day based
on increased incidence of
proteinaceous casts in the kidneys,
and statistically significant increases
in the absolute weights of the thyroid and adrenal glands in males.
Ref:
Sept 24, 2004. Lactofen. Pesticide Tolerance. Final Rule. Federal
Register.
http://www.fluorideaction.org/pesticides/lactofen.fr.sept.24.2004.htm
Eye
(click
on for all fluorinated pesticides)
At 37.5 mg/kg/day,
there was also an increased incidence of cataracts
and renal pigmentation. Based on the LOEL, an oral RfD of 0.002
mg/kg/day was derived.
Ref:
USEPA/OPP. Support Document for the Addition of Chemicals from
Federal Insecticide, Fungicide, Rodenticide Act (FIFRA) Active
Ingredients to EPCRA Section 313. U. S. Environmental Protection
Agency, Washington, DC (1993). As cited by US EPA in: Federal
Register: January 12, 1994. Part IV. 40 CFR Part 372. Addition
of Certain Chemicals; Toxic Chemical Release Reporting; Community
Right-to-Know; Proposed Rule.
Kidney
(click
on for all fluorinated pesticides)
Chronic toxicity--dogs. NOAEL = 0.79 mg/kg/day. LOAEL = 3.96
mg/kg/day based on increased incidence of proteinaceous
casts in the kidneys, and statistically
significant increases in the absolute weights of the thyroid and
adrenal glands in males.
Ref:
Sept 24, 2004. Lactofen. Pesticide Tolerance. Final Rule. Federal
Register.
http://www.fluorideaction.org/pesticides/lactofen.fr.sept.24.2004.htm
Results of several subchronic and chronic studies indicated the
liver and kidney as target organs
for lactofen. Increased absolute and relative liver weight and
hepatocytomegaly (the LOEL was 1.5 mg/kg/day; the NOEL was not
determined) were observed in male mice fed lactofen for 78 weeks.
At 37.5 mg/kg/day, there was also an increased incidence of cataracts
and renal pigmentation. Based on the LOEL, an oral RfD of 0.002
mg/kg/day was derived. Renal dysfunction
and decreased hemoglobin and hematocrit levels and red blood cell
counts (the LOEL was 25/75 mg/ kg/day; the NOEL was 5 mg/kg/day)
were observed in a 1-year feeding study in dogs. Increased renal
and hepatic pigmentation (the LOEL was 50 mg/kg/day; the
NOEL was 25 mg/kg/day) were noted in a 2-year feeding study in
rats. In a 90-day mouse study, increased alkaline phosphatase,
serum glutamate oxaloacetate transaminase (SGOT), and serum gleutanic
pyruvic transaminase (SGPT) activities, increased liver weight,
hepatic necrosis, biliary hyperplasia,
decreased hematocrit and hemoglobin levels and red blood cell
counts, extramedullary hematopoiesis, and
kidney nephrosis and fibrosis (the LOEL was 26 mg/kg/day;
the NOEL was not determined) were seen. Decreased hemoglobin and
hematocrit levels, decreased red blood cell counts, and brown
pigment in the kidney and liver (the LOEL was 50 mg/kg/day) were
noted in a 90-day feeding study in rats. EPA believes that there
is sufficient evidence for listing lactofen on EPCRA section 313
pursuant to EPCRA section 313(d)(2)(B) based on the available
carcinogenicity data and hepatic,
renal, and hematological toxicity data for this chemical.
Ref: USEPA/OPP. Support Document for the
Addition of Chemicals from Federal Insecticide, Fungicide, Rodenticide
Act (FIFRA) Active Ingredients to EPCRA Section 313. U. S. Environmental
Protection Agency, Washington, DC (1993). As cited by US EPA in:
Federal
Register: January 12, 1994. Part IV. 40 CFR Part 372. Addition
of Certain Chemicals; Toxic Chemical Release Reporting; Community
Right-to-Know; Proposed Rule.
Chronic Toxicity- A complete chronic data base supported by appropriate
subchronic studies for lactofen is available to the Agency. Lactofen
Technical causes adverse health effects when administered to animals
for extended periods of time. These effects include proliferative
changes in the liver, spleen, and kidney;
hematological changes; and blood biochemistry changes. Based on
the Lowest Effect Level (LEL) of 1.5 mg/ kg/day in the 18-month
mouse feeding study and an uncertainty factor of 1,000, a reference
dose (RfD) of 0.002 mg/kg/day has been established for lactofen.
An uncertainty factor of 1,000 was used since a clear NOEL was
not established.
-- Reproduction-- Rats. Groups of male and female rats were fed
0, 50, 500 or 2,000 ppm of Lactofen Technical continuously in
their diets for 2-generations. Adult systemic toxicity (mortality,
reduced body weight, increased liver and spleen weight, decreased
kidney weight and histological changes
in the liver and testes) was observed at levels of 500 ppm and
greater.
-- Chronic/carcinogenicity feeding study-- Rat-- 24-month. In
a 2- year chronic feeding/oncogenicity study of Lactofen Technical
in rats at dosages of 0, 500, 1,000 and 2,000 ppm in the diet,
an increase in liver neoplastic nodules and foci of cellular alteration
was observed in both sexes at 2,000 ppm. The NOEL for systemic
toxicity is 500 ppm based on kidney
and liver pigmentation.
Ref: Federal Register: February 25, 1998
[Page 9532-9540]. Notice of Filing of Pesticide Petition.
http://www.fluoridealert.org/pesticides/Lactofen.FR.Feb.1998.htm
Liver
(click
on for all fluorinated pesticides)
Likely
to be Carcinogenic to Humans at High Doses. Not
Likely to be Carcinogenic to Humans at Low Doses. Hepatocellular
carcinomas
(M); Hepatocellular adenomas & carcinomas (M & F); CD-mice.
Liver neoplastic nodules; Sprague-Dawley rats (M & F). MOE
approach should be used for estimating human cancer risk, using
a NOAEL of 2 ppm (0.3 mg/kg/day)
Ref: April
26, 2006 . Chemicals Evaluated for Carcinogenic Potential by the
Office of Pesticide Programs. From: Jess Rowland, Chief Science
Information Management Branch Health Effect Division (7509C) Office
of Pesticide Programs, USEPA.
http://www.fluorideaction.org/pesticides/pesticides.cancer.potential.2006.pdf
Lactofen acts via a peroxisome proliferation mechanism of action.
Likely to be carcinogenic to humans at high enough doses to cause
these biochemical and histopathological effects (peroxisome
proliferation) in the livers of rodents but unlikely to
be carcinogenic at doses below those causing these changes. Lactofen
is considered to be a threshold carcinogen. NOAEL = 0.3
mg/kg/day based on increased activities
of liver enzymes and increased incidence of liver histopathological
findings at the LOAEL of 1.5 mg/kg/day.
Ref:
Sept 24, 2004. Lactofen. Pesticide Tolerance. Final Rule. Federal
Register.
http://www.fluorideaction.org/pesticides/lactofen.fr.sept.24.2004.htm
Lactofen meets the
criteria of an EPA Group B2 compound, i.e., a probable human carcinogen.
This conclusion was based on an increased incidence of hepatocellular
carcinomas in males and combined incidence of hepatocellular
adenomas and carcinomas in both sexes of CD-1 mice following
dietary administration of lactofen. In CD rats, there was increased
incidence of liver neoplastic nodules in
both sexes. Four structurally similar chemicals, acifluorfen,
nitrofen, oxyfluorfen, and fomesafen, all produced hepatocellular
tumors in rodents. Results of several subchronic and chronic studies
indicated the liver and kidney as
target organs for lactofen. Increased absolute and relative liver
weight and hepatocytomegaly (the LOEL was 1.5 mg/kg/day;
the NOEL was not determined) were observed in
male mice fed lactofen for 78 weeks. At 37.5 mg/kg/day,
there was also an increased incidence of cataracts and renal pigmentation.
Based on the LOEL, an oral RfD of 0.002 mg/kg/day was derived.
Renal dysfunction and decreased hemoglobin and hematocrit levels
and red blood cell counts (the LOEL was 25/75 mg/ kg/day; the
NOEL was 5 mg/kg/day) were observed in a 1-year feeding study
in dogs. Increased renal and hepatic pigmentation (the LOEL was
50 mg/kg/day; the NOEL was 25 mg/kg/day) were noted in a 2-year
feeding study in rats. In a 90-day mouse study, increased alkaline
phosphatase, serum glutamate oxaloacetate transaminase (SGOT),
and serum gleutanic pyruvic transaminase (SGPT) activities, increased
liver weight, hepatic necrosis, biliary hyperplasia, decreased
hematocrit and hemoglobin levels and red blood cell counts, extramedullary
hematopoiesis, and kidney nephrosis and fibrosis (the LOEL was
26 mg/kg/day; the NOEL was not determined) were seen. Decreased
hemoglobin and hematocrit levels, decreased red blood cell counts,
and brown pigment in the kidney and liver
(the LOEL was 50 mg/kg/day) were noted in a 90-day feeding study
in rats. EPA believes that there is sufficient evidence for listing
lactofen on EPCRA section 313 pursuant to EPCRA section 313(d)(2)(B)
based on the available carcinogenicity data and hepatic,
renal, and hematological toxicity data for this chemical.
Ref:
USEPA/OPP. Support Document for the Addition of Chemicals from
Federal Insecticide, Fungicide, Rodenticide Act (FIFRA) Active
Ingredients to EPCRA Section 313. U. S. Environmental Protection
Agency, Washington, DC (1993). As cited by US EPA in: Federal
Register: January 12, 1994. Part IV. 40 CFR Part 372. Addition
of Certain Chemicals; Toxic Chemical Release Reporting; Community
Right-to-Know; Proposed Rule.
Chronic/carcinogenicity
feeding study-- Mouse-- 24-month. In a dietary 18-month oncogenicity
study in mice at dosages of 10, 50 and 250 ppm Lactofen Technical,
an increase in liver adenomas and
carcinomas, cataracts and liver pigmentation
was observed at 250 ppm. The lowest dose, 10 ppm, was the LOEL
based on increased liver weight and hepatocytomegaly.
-- Chronic/carcinogenicity feeding study-- Rat-- 24-month. In
a 2- year chronic feeding/oncogenicity study of Lactofen Technical
in rats at dosages of 0, 500, 1,000 and 2,000 ppm in the diet,
an increase in liver neoplastic nodules
and foci of cellular alteration was observed in both sexes at
2,000 ppm. The NOEL for systemic toxicity is 500 ppm based on
kidney and liver pigmentation.
-- Carcinogenicity. The Toxicology Branch Peer Review Committee
has determined that lactofen meets the criterion for a B2 (possible
human) carcinogen since it caused an increase in liver
tumors (adenomas and/or carcinomas) in two species. Based
on the mouse oncogenicity study, a human upper-bound potency estimate
(Q1*) was calculated as 0.17 (mg/kg/day).
-- More importantly, evidence summarized above suggest that carcinogenic
effects observed in rodent liver related to long term lactofen
consumption are attributable to peroxisomal
proliferation as opposed to a direct genotoxic effect.
This mechanism of action would more appropriately be regulated
as a threshold effect (similar to RfD comparisons) as opposed
to a non-threshold effect with a quantitative potency factor derived
from low dose extrapolations. This change in the hazard assessment
process for lactofen would have a profound effect on the exposure
and risk assessments for this chemical.
Ref: Federal Register: February 25, 1998
[Page 9532-9540]. Notice of Filing of Pesticide Petition.
http://www.fluoridealert.org/pesticides/Lactofen.FR.Feb.1998.htm
Reproductive
(click
on for all fluorinated pesticides)
Reproduction
and fertility effects
Parental/Systemic NOAEL = 2.6 mg/kg/day.
Parental/Systemic LOAEL = 26.2 mg/kg/day based on mortality and
decreased male fertility.
Reproductive NOAEL = 2.6 mg/kg/day.
Reproductive LOAEL = 26.2 mg/kg/day based on decreased
male fertility.
Ref: Sept 24, 2004.
Lactofen. Pesticide Tolerance. Final Rule. Federal Register.
http://www.fluorideaction.org/pesticides/lactofen.fr.sept.24.2004.htm
Spleen
(click
on for all fluorinated pesticides)
--
Subchronic feeding-- Mice-- 3-month. Groups of Male and female
mice were fed diets containing Lactofen Technical at concentrations
of 0, 40, 200, 1,000, 5,000, and 10,000 for 13-weeks. At week
5, the dosage of the 40 ppm groups was increased to 2,000 ppm.
Treatment related mortality occurred at dosages above 1,000 ppm.
The LOEL was 200 ppm based on: increased WBC; decreased hematocrit,
hemoglobin and RBC; increased alkaline phosphatase, SGOT, SGPT,
cholesterol and total serum protein levels; increased weights
or enlargement of the spleen, liver, adrenals, heart and kidney;
histopathological changes of the liver, kidney, thymus,
spleen, ovaries and testes. In general, effects were slight
in the 200 ppm groups, and moderate to severe in the 1,000 ppm
groups.
-- Reproduction-- Rats. Groups of male and female rats were fed
0, 50, 500 or 2,000 ppm of Lactofen Technical continuously in
their diets for 2-generations. Adult systemic toxicity (mortality,
reduced body weight, increased liver and
spleen weight, decreased kidney weight and histological
changes in the liver and testes) was observed
at levels of 500 ppm and greater.
Ref: Federal Register: February 25, 1998
[Page 9532-9540]. Notice of Filing of Pesticide Petition.
http://www.fluoridealert.org/pesticides/Lactofen.FR.Feb.1998.htm
| Environmental
(click on for
all fluorinated pesticides)
Phototoxic
Pesticide.
Light-dependent peroxidizing herbicides
(LDPHs). US EPA identified the organofluorine herbicides
Acifluorfen, Azafenidin, Carfentrazone-ethyl, Flumiclorac-penty,
Flumioxazin, Fluthiacet-methyl, Fomesafen, Lactofen,
Oxadiargyl, Oxadiazon, Oxyfluorfen,
Sulfentrazone, Thidiazimin as phototoxic
pesticides that act by inhibiting protoporphyringen
oxidase in the heme and chlorophyll biosynthetic pathway.
[10 out of the 13 pesticides that EPA identified are organofluorines].
SEE http://www.fluoridealert.org/pesticides/PHOTOTOXICITY.PAGE.htm
Ref: December 11, 2001 - US EPA. Revised
Environmental Fate and Effects Division Preliminary Risk
Assessment for the Oxyfluorfen Reregistration Eligibility
Decision Document - also at: http://www.epa.gov/oppsrrd1/reregistration/oxyfluorfen/oxyefedchap.pdf
|
|

