|
Return to Fluazinam
Index Page
Activity:
Fungicide
(2,6-Dinitroaniline)
Structure:
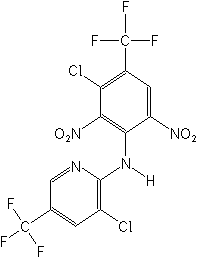
Adverse
Effects:
Body
Weight Decrease
Bone (including
Cleft Palate)
Brain
Cancer-
Suggestive evidence: THYROID,
LIVER
Cholesterol
Dermal
Endocrine: Testicular
Endocrine: Thymus
Endocrine:
Thyroid
Endocrine: Uterus
Eye
Liver
Lung
Pancreas
Spinal Cord
Stomach
Thyroid
Environmental
•
As of February 16, 2005, this fungicide is permitted in
or on 3 food commodities in the United States - see
list at bottom of page.
•
In subchronic and chronic toxicity studies, fluazinam targeted
the following organs: liver, lung, uterus, testes, pancreas,
thymus, thyroid, stomach, eyes and brain. Generalized toxicity
was observed in rats, mice and dogs as decreases in body
weight, body-weight gain, food consumption and/or food efficiency.
Liver toxicity was evident in most studies including increased
size and weight, fatty changes, pallor, as well as hepatocyte
hypertrophy, necrosis and apoptosis. Thyroid toxicity was
less common, but included follicular hyperplasia and cystic
thyroid follicles. Endocrine-related effects included small
and/or flaccid testes, testicular tubular atrophy, pancreatic
exocrine atrophy and thymic hyperplasia.
Ref:
Canada: Regulatory Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October
27, 2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
|
Body
Weight Decrease (click
on for all fluorinated pesticides)
In subchronic and chronic
toxicity studies, fluazinam targeted the following organs: liver,
lung, uterus, testes, pancreas, thymus, thyroid, stomach, eyes
and brain. Generalized toxicity was observed
in rats, mice and dogs as decreases in body weight, body-weight
gain, food consumption and/or food efficiency.
Ref:
Canada: Regulatory
Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October 27,
2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
Prenatal developmental
toxicity -rats. LOAEL = 250 mg/kg/day based on decreased
fetal body weights and placental weights,
increased facial/cleft palates, diaphragmatic hernia, and delayed
ossification in several bone types, greenish amniotic fluid and
possible increased late resorptions and postimplantation loss...
Prenatal developmental toxicity -rabbits. LOAEL = 12 mg/kg/day
based on an increase in total litter resorptions and possible
fetal skeletal abnormalities.
Ref: Federal Register. September 7, 2001.
Fluazinam; Pesticide Tolerance. Final Rule. http://www.fluoridealert.org/pesticides/Fluazinam.FR.Sept.7.2001.htm
-- Prenatal developmental
toxicity rats Maternal. NOAEL = 50 mg/kg/day LOAEL = 250 mg/kg/day
based on decreased body weight gain and food consumption and increased
water consumption and urogenital staining Developmental NOAEL
= 50 mg/kg/day LOAEL = 250 mg/kg/day based on decreased
fetal body weights and placental weights, increased facial/cleft
palates, diaphragmatic hernia, and delayed ossification in several
bone types, greenish amniotic fluid and possible increased late
resorptions and postimplantation loss
-- Reproduction and fertility effects rats. Parental/Systemic
NOAEL = 1.9 mg/kg/day LOAEL = 9.7 mg/kg/day based on liver pathology
in F1 males Reproductive NOAEL = 10.6 mg/kg/day LOAEL = 53.6 mg/kg/day
based on decreased number of implantation sites and decreased
litter sizes to day 4 post-partum for F1 females (F2 litters).
Offspring NOAEL = 8.4 mg/kg/day LOAEL = 42.1 mg/kg/day based on
reduced F1 and F2 pup body weight gains
during lactation.
-- Specicial study. 4-Week dietary (range-finding) rats. NOAEL
= M: 5.1 mg/kg/day; F: 5.3 mg/kg/day LOAEL = M: 26.4 mg/kg/day;
F: 25.9 mg/kg/day based on decreased body
weight gain and food consumption,
increased serum phospholipids, increased total cholesterol, increased
relative liver weights, and liver histopathology.
-- Specicial study. 4-Week dietary (Range-finding) mice NOAEL
= M: 7.6 mg/kg/day; F: 8.2 mg/kg/day LOAEL = M: 36 mg/kg/day;
F: 43 mg/kg/day based on decreased body
weight gain, increased serum glucose, increased kidney
weights.
Ecological Effects
-- a. Aquatic (Acute/Chronic Hazard Summary) Fluazinam is considered
to be very highly toxic to highly toxic
to fish (freshwater and estuarine/marine) on an acute basis (LC50
= 0.036 - 0.11 ppm). Chronic freshwater NOAEC/LOAEC values were
calculated at 0.0053 - 0.00069 ppm and 0.010 - 0.014 ppm, respectively,
with larval survival, reduced number of spawns, and growth as
the endpoints affected. Acute toxicity values for aquatic invertebrates
suggest that fluazinam is highly toxic to freshwater invertebrates
(Daphnia EC50 = 0.18 - 0.22 ppm) and very highly toxic to estuarine/marine
invertebrates (oyster EC50 = 0.0047 and mysid shrimp EC50 = 0.039
ppm ). Chronic toxicity to invertebrates are only represented
through the Daphnia magna life cycle where the NOAEC was calculated
at 0.068 ppm and the LOAEC at 0.140 ppm.. The
endpoints affected for this study were reproductive (reduced number
of young per female) and growth effects. No acceptable
data have been submitted to assess the chronic effects of fluazinam
to estuarine/marine fish or invertebrates. An estuarine/marine
fish life-stage toxicity test (Guideline 72-4a) and an estuarine/marine
invertebrate life-cycle toxicity test (Guideline 72-4b) are required
to fulfill these requirements.
-- d. Risk to Avian Species (Acute/Chronic) Although acute exposure
should result in minimal toxic effects to birds, the
risk assessment suggests that the proposed uses can cause chronic
(reduced growth in young) effects in birds. RQ values
were calculated for exposure to peanuts (maximum EECs RQ = 1.0
- 1.8 and 56 day average EECs RQ = 1.1 ppm) and potatoes (maximum
EECs RQ = 1.0 - 1.5 and 56 day average(RQ = 1).
Ref: US EPA Pesticide Fact Sheet. Fluazinam.
August 10, 2001.
http://www.epa.gov/opprd001/factsheets/fluazinam.pdf
Bone
(including Cleft Palate)
(click on for all fluorinated pesticides)
-- Prenatal
developmental toxicity rats. Maternal NOAEL = 50 mg/kg/day LOAEL
= 250 mg/kg/ day based on decreased body
weight gain and food consumption and increased water consumption
and urogenital staining Developmental NOAEL = 50 mg/kg/ day LOAEL
= 250 mg/kg/ day based on decreased fetal body weights and placental
weights, increased
facial/ cleft palates,
diaphragmatic hernia, and delayed ossification
in several bone types, greenish amniotic fluid and possible
increased late resorptions and postimplant
-- Prenatal developmental toxicity rabbits. Maternal NOAEL = 4
mg/kg/day LOAEL = 7 mg/kg/ day based on decreased food consumption
and increased liver histopathology. Developmental NOAEL = 7 mg/kg/
day LOAEL = 12 mg/kg/ day based on an increase in total litter
resorptions and possible fetal skeletal
abnormalities
Ref: Federal Register: April 18,
2002. Fluazinam; Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.Apr.18.2002.htm
Developmental. 98.5%,
NZW Rabbits, 0, 2, 4, 7, 12 mg/kg/d, 16Ð18 pregnant females/dose
Maternal NOAEL 4 mg/kg/d Maternal LOAEL 7 mg/kg/d Developmental
NOAEL 7 mg/kg/d Developmental LOAEL 12 mg/kg/d Qualitative sensitivity
of young. 7 mg/kg/d 9 Food consumption. 8 Liver histopath (cellular
hypertrophy, single cell necrosis, binucleate hepatocytes, brown
pigment deposition, apoptosis). 12 mg/kg/d 9 bwg, food consumption.
8 Liver histopath (cellular hypertrophy, single cell necrosis,
binucleate hepatocytes, brown pigment deposition, apoptosis).
8 Incidence total litter resorptions. 8 Incidence placental anomalies.
8 Skeletal abnormalities (slight) (kinked
tail tip, fused or incompletely ossified sternebrae, abnormalities
of head bones).
Ref:
Canada: Regulatory Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October 27,
2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
Brain
(click on for all fluorinated pesticides)
-- In subchronic and
chronic toxicity studies, fluazinam targeted the following organs:
liver, lung, uterus, testes, pancreas, thymus, thyroid, stomach,
eyes and brain...
-- In acute and subchronic neurotoxicity studies, treatment with
fluazinam resulted in marked neuropathology
in the form of vacuolation of the white matter of the brain.
Special
studies have been submitted that show this effect is not due to
fluazinam itself, but rather to a manufacturing impurity.
In the subchronic study, generalized toxicity was observed as
decreased body weight and food efficiency. A developmental neurotoxicity
study was suggested upon preliminary review of the data and the
applicant submitted a rationale. Upon full review of the dataset,
a developmental neurotoxicity study is a
requirement for full registration. Special
studies were generated to determine the cause of the white matter
vacuolation. All
nine impurities were tested at doses that reflected their relative
content in the technical product. Only one
impurity was found to produce the vacuolation effects and all
subsequent special studies focus on this chemical. The vacuolation
was determined to be in the myelin sheaths surrounding axons in
the white matter. Dogs, mice and rats were found to have comparable
results when tested in similar manners. Older animals were found
to be more susceptible than the young. All vacuolation
effects were found to be reversible. Neurotoxic findings were
common at high doses and included decreased motor activity, partial
paralysis and ataxia. This effect was observed at high doses (LOAEL
was 50 mg/kg/d) when the dose level of impurity no. 5 was sufficiently
high to cause these effects. Therefore, the
presence of this impurity will be limited to 0.1% of the technical
active.
-- Determination of acceptable daily intake
(ADI). The recommended ADI for fluazinam is 0.0011 mg/kg bw/day.
The chronic toxicity and oncogenicity study in mice was considered
the most appropriate study to assess chronic dietary exposure.
The no observed adverse effect level (NOAEL) was 1.1 mg/kg bw/day,
based on increased incidences of brown pigmented macrophages in
the liver, increased incidences of eosinophilic vacuolated hepatocytes
in males and increased liver weights relative to body weights.
The standard uncertainty factor of 100-fold is applied to account
for intraspecies and interspecies variability, an additional 10-fold
safety factor is recommended to firstly, protect for endocrine-related
effects and secondly, to account for the lack of a developmental
neurotoxicity (DNT) study. This provides
margins of safety of 9100 to the NOEL for white matter vacuolation
and 6350 to the NOEL for developmental effects in the rabbit developmental
study.
-- The USEPA has chosen an ADI of 0.00367 mg/kg bw/d based on
a 2-year carcinogenicity study in mice (1.1 mg/kg bw/d with 100-fold
uncertainty factor (UF) and three-fold Food Quality Protection
Act (FQPA) safety factor.) They have also
requested a developmental neurotoxicity study.
Ref:
Canada: Regulatory
Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October 27,
2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
90-Day oral toxicity
dogs NOAEL = 10 mg/kg/day LOAEL = 100 mg/kg/day based on retinal
effects, increased relative liver weight, liver histopathology
and possible increased serum alkaline phosphatase in females and
possible marginal vacuolation of the cerebral
white matter (equivocal)...
Carcinogenicity mice NOAEL = Males:<126 mg/kg/day, Females: <162
mg/kg/day LOAEL = Males: 126 mg/kg/day; Females: 162 mg/kg/day
based on increased liver weights and liver and brain
histopathology in both sexes
Special study: 4-Week dietary (Range-finding) mice NOAEL = not
identified (Males; <555 mg/kg/day; Females: <658 mg/ kg/day) LOAEL
= Males: 555 mg/kg/day; Females: 658 mg/kg/day based on
vacuolation of white matter in brain, increased liver weights,
histopathology in liver...
Eight special mechanistic studies to assess the
CNS white matter vacuolation: White matter vacuolation in the
CNS of mice, rats, and dogs was found to be due to Impurity-5.
Ref: Federal Register. September 7, 2001.
Fluazinam; Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.Sept.7.2001.htm
A second oncogenicity study in mice was conducted at 1,000,
3,000 and 7,000 ppm to ensure that an maximum tolerance dose (MTD)
was studied. Findings included increased female mortality, reduced
body weight gains, increased brain weights
and/or liver weights. An impurity in the test material used in
this study resulted in vacuolation of the
white matter of the brain and cervical spinal cord in treated
animals. A statistically significant higher incidence of hepatocellular
adenomas was observed in the 3,000 ppm dose males. Hepatocellular
adenomas are common tumors in male mice. There was no dose relationship
in the induction of the adenoma and no increase in hepatocellular
carcinomas. It was concluded that fluazinam is not carcinogenic
in the mouse.
Ref: Federal Register: December 6, 2000
[Page 76253-76258].
http://www.fluoridealert.org/pesticides/Fluazinam.FR.December.2000.htm
Of particular concern
was a neurotoxic lesion described as vacuolation
of the white matter of the brain and sometimes cervical
spinal cord which was initially observed in long-term (1-2 year)
chronic studies in mice and dogs and later, upon careful re-examination
of the central nervous system (CNS), also in shorter-term (4-week
to 90-day) subchronic studies. Although this lesion was also observed
in control animals, the increased incidence and/or severity of
the lesion in test animals was clearly treatment-related and dose-related.
Further investigation of this finding in a series of special studies
demonstrated the same lesion could also be induced in rats. In
the special studies, the following were also determined.
• Fluazinam, per se, was not responsible for the induction
of this lesion. Evaluation of the effects of impurities present
in technical grade fluazinam revealed that one single impurity,
Impurity-5, was solely responsible for the induction of this lesion.
• No significant differences in susceptibility or in incidence
or severity of vacuolation of the white matter of the CNS were
observed between species (mice, dogs, or rats). Similarly, no
significant differences were attributed to sex.
• Electron microscopy of the white
matter (cerebellum) of mice treated with technical grade fluazinam
indicated that treatment-related effects were confined to the
myelin sheaths. Large vacuoles were observed in the intramyelin
sheaths due to the accumulation of fluid between the sheaths.
The nucleus and mitochondria in the oligodendroglia were observed
to remain intact, suggesting no damage to these cells.
• White matter vacuolation in the CNS was reversible. The
myelin sheaths appeared to recover completely during a recovery
period of up to 56 days.
• There appears to be a non-linear dose-response with a clear
threshold below which no effect occurs. It was concluded that
a LOAEL of 0.1 mg/kg/day and a NOAEL of 0.02 mg/kg/day for CNS
effects could be established for Impurity-5.
-- At the current maximum concentration of Impurity-5 in technical
grade fluazinam of 0.1%, the NOAEL for CNS effects of 0.02 mg/kg/day
for Impurity-5 is equivalent to a NOAEL for CNS effects of 20
mg/kg/day for technical grade fluazinam. The NOAEL of 20 mg/kg/day
for CNS effects for technical grade fluazinam is comparable to
the NOAEL for chronic effects for technical grade fluazinam of
1.1 mg/kg/day used to establish the chronic RfD for fluazinam.
Therefore, based on a consideration of all the available data
and information relating to this treatment-related neurotoxic
lesion, it was concluded that the chronic dietary RfD of 0.011
mg/kg/day for Òall populationsÓ, including infants and children,
is protective of the CNS effects caused by the presence of Impurity-5
in technical-grade fluazinam.
Ref: US EPA Pesticide Fact Sheet. Fluazinam.
August 10, 2001. http://www.epa.gov/opprd001/factsheets/fluazinam.pdf
SPECIAL/IMPURITIES
-- ComparativeÑ susceptibility to neurotoxicity.
97.0% Impurity-5, 2.0 mg/kg/d, 5
Crj:CD (ICR) mice, 5 Crj:CD (SD) rats, 3 beagles, all male 9 Spontaneous
motor activity in rats and mice. 9 Mean body weight in mice and
rats. 8 Brain weights in mice and rats.
Vacuolation of white matter and swelling of the brain in all treated
animals. Dogs appear to be slightly less susceptible.
-- ComparativeÑ brain sensitivity rats and
mice. 99.5% Impurity-5, 0,
0.5 mg/kg, 5 Crj:CD-1 mice, 5 Crj:CD(SD)
SPF/VAF rats, all female. Incidence
and severity of white matter vacuolation in brains of mice and
rats were similar. Brain weights were comparable to controls.
-- ComparativeÑ brain sensitivity 3- and
10-week old rats and miceÑ14 days. 99.5%
Impurity-5, 0, 0.5 mg/kg/d, 10 Crj:CD-1 (ICR) SPF/VAF mice
and Crj:CD(SD) SPF/VAF rats,
all male, half 3 weeks old, the other half 10 weeks old
Edema in brains of 1 rat and 2 mice (10
weeks old). Vacuolation in white matter of brains similar between
species of same age, but more severe in older animals compared
to younger.
-- Effect on brain/optic nerves 99.5%.
Impurity-5, 2.5
mg/kg, Crj:CD-1(ICR) mice, 5 males/group, aged 3, 5, 8, 10, 12,
16, 20, 24 weeks All treated mice except
3 week olds had vacuolation of the optic nerve. The effect
was less severe than in the brain. The eyes themselves were normal.
Ages 3, 5, 8 weeks. Trace vacuolation of
white matter in brains Ages 10, 12, 16, 20, 24
weeks Trace to minimal vacuolation of white matter in brains.
-- Various impuritiesÑ effect on brains.
96Ð100% Impurities 1 through 9, doses
correspond to 5000 mg/kg dose of technical, 5 male Crj:CD-1 mice
(Impurity-5 was dosed at 5 mg/kg) Only Impurity-5 was toxic. Coarse
fur, paralysis of hind legs, staggering gait, sedation, moribund.
9 Mean body weight. 8 Brain weight, edema.
Vacuolation of white matter.
-- Recommended
ADI.
0.0011 mg/kg bw/d based on 2 year carcinogenicity in mice (1.1
mg/kg/d with 100- fold UF, three-fold SF for endocrine-related
effects (testicular atrophy, pancreatic exocrine atrophy), and
three-fold for lack of DNT). MOS for other critical endpoint(s)
White matter vacuolation/Neurotox NOEL for white matter vacuolation
was 10 mg/kg/d in chronic dog study (equivalent to 0.02 mg/kg/d
Impurity-5). MOS = 9100. Tumours NOEL for tumours was 3.8
mg/kg bw/d in 2-year rat study. MOS = 3450. Developmental effects
NOAEL for developmental effects was 7 mg/kg bw/d in developmental
rabbit study. MOS = 6350
[ MOS = Margin of Safety
]
Ref:
Canada: Regulatory
Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October 27,
2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
Cancer:
LIVER, THYROID
(click on for all fluorinated pesticides)
Suggestive
Evidence of Carcinogenicity to Humans.
An increase in thyroid gland follicular
cell tumors in male rats, and an increased incidence of hepatocellular
tumors observed in the male mice was treatment-related.
Ref: April
26, 2006 . Chemicals Evaluated for Carcinogenic Potential by the
Office of Pesticide Programs. From: Jess Rowland, Chief Science
Information Management Branch Health Effect Division (7509C) Office
of Pesticide Programs, USEPA.
http://www.fluorideaction.org/pesticides/pesticides.cancer.potential.2006.pdf
-- Carcinogenicity
mice NOAEL = Males:1.1 mg/kg/day; Females: 1.2 mg/kg/day [[Page
46732]] LOAEL = Males: 10.7 mg/kg/day; Females: 11.7 mg/kg/day
based on increased incidences of brown macrophages in the
liver of both sexes, eosinophilic vacuolated hepatocytes in males,
and increased liver weight in females.
Clear evidence of carcinogenicity (hepatocellular tumors) in male
mice, but not in females.
-- Combined chronictoxicity/ NOAEL = Males: 0.38 mg/kg/day; carcinogenicity
rats Females: 0.47 mg/kg/day LOAEL = Males: 3.8 mg/kg/day; Females:
4.9 mg/kg/day based on liver toxicity
in both sexes, pancreatic exocrine atrophy in females and testicular
atrophy in males. Some evidence of carcinogenicity
(thyroid gland follicular cell tumors) in male rats, but not in
females.
-- Carcinogenicity mice NOAEL = Males:<126 mg/kg/day, Females:
<162 mg/kg/day LOAEL = Males: 126 mg/kg/day;
Females: 162 mg/kg/day based on increased liver weights and liver
and brain histopathology in both sexes Equivocal/some
evidence of carcinogenicity (hepatocellular tumors) in male mice,
but not in females,
Ref: Federal Register. September 7, 2001.
Fluazinam; Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.Sept.7.2001.htm
-- Cancer (oral, dermal, inhalation). `Suggestive evidence of
carcinogenicity, but not sufficient to assess human carcinogenic
potential'' 2 . Quantification of human cancer risk not required.
2 Increases in thyroid gland follicular
cell tumors in male rats; increases
in hepatocellular (liver) tumors in male mice.2 (Ref 2:
2Cancer Assessment Document - Evaluation of the Carcinogenic Potential
of Fluazinam, March 29, 2001, HED Doc. No. 014512.)
-- Cancer. Since fluazinam has been classified as ``Suggestive
evidence of carcinogenicity, but not sufficient to assess human
carcinogenic potential,'' an exposure assessment was not performed.
Ref: Federal Register: April 18, 2002. Fluazinam;
Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.Apr.18.2002.htm
Cancer:
Some evidence of carcinogenicity in male rats - THYROID
(click on for all fluorinated pesticides)
-- Combined chronictoxicity/
NOAEL = Males: 0.38 mg/kg/day; carcinogenicity rats Females: 0.47
mg/kg/day LOAEL = Males: 3.8 mg/kg/day; Females: 4.9 mg/kg/day
based on liver toxicity in both sexes,
pancreatic exocrine atrophy in females and testicular atrophy
in males. Some evidence of carcinogenicity
(thyroid gland follicular cell tumors) in male rats, but not in
females.
Ref: Federal Register. September 7, 2001.
Fluazinam; Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.Sept.7.2001.htm
-- Cancer (oral, dermal, inhalation). `Suggestive evidence of
carcinogenicity, but not sufficient to assess human carcinogenic
potential'' 2 . Quantification of human cancer risk not required.
2 Increases in thyroid gland follicular
cell tumors in male rats; increases in hepatocellular
(liver) tumors in male mice.2
(Ref 2: 2Cancer Assessment Document - Evaluation of the Carcinogenic
Potential of Fluazinam, March 29, 2001, HED Doc. No. 014512.)
-- Cancer. Since fluazinam has been classified as ``Suggestive
evidence of carcinogenicity, but not sufficient to assess human
carcinogenic potential,'' an exposure assessment was not performed.
Ref: Federal Register: April 18, 2002. Fluazinam;
Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.Apr.18.2002.htm
Cholesterol
(click on for all fluorinated pesticides)
-- 21-Day dermal toxicity
rats: increased AST and cholesterol
levels in clinical chemistry determinations (males).
Special studies: 4-Week dietary (Range-finding) rats- decreased
body weight gain and food consumption, increased serum phospholipids,
increased total cholesterol,
increased relative liver weights, and liver histopathology.
-- 4-Week dietary (Range-finding) mice- decreased body weight
gain, increased serum glucose, increased
kidney weights.
-- 4-Week dietary (Range-finding) rats. NOAEL = Males: 5.1 mg/kg/day;
Females: 5.3 mg/ kg/day LOAEL = Males: 26.4 mg/kg/day; Females:
25.9 mg/ kg/day based on decreased body
weight gain and food consumption, increased serum phospholipids,
increased total cholesterol, increased
relative liver weights, and liver histopathology.
Ref: Federal Register: April 18, 2002 [Page
19120-19130]. Fluazinam; Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.Apr.18.2002.htm
Dermal
(click on for all
fluorinated pesticides)
Abstract:
In spring 1992,
several farmers in the western part of The Netherlands developed
dermatitis on their hands, forearms and
face. In some, the legs, trunk and genitals were also affected.
Complaints ranged from a mildly itchy, papular rash to a painful,
weeping and blistering dermatitis. Medical aid was needed
by 5/9 of them. Some of the farmers grew seed potatoes, the others
cultivated lilies. All of them had in common
that their complaints emerged after repeated application of a
new fungicide over several weeks. The fungicide was Shirlan,
with fluazinam as its active ingredient. 9 farmers were patch
tested with a concentration range of the whole formulation (aq.)
and of the active ingredient (pet.). In
7 of 9 farmers, positive patch tests were scored with both the
whole formulation (down to 0.01% aq.) and fluazinam itself (down
to 0.1% pet.). Patch tests in consecutive control patients
(n = 10) were all negative. As it was impossible to substitute
fluazinam as the active ingredient, farmers are now supplied with
detailed information as to how to avoid skin contact as much as
feasible.
Ref: Contact Dermatitis 1995 Mar;32(3):160-2;
Allergic
contact dermatitis from the newly introduced fungicide fluazinam;
by van Ginkel CJ, Sabapathy NN.
Endocrine
Pancreas (click
on for all fluorinated pesticides)
-- In subchronic and
chronic toxicity studies, fluazinam targeted the following organs:
liver, lung, uterus, testes, pancreas,
thymus, thyroid, stomach, eyes and brain... Endocrine-related
effects included small and/or flaccid testes, testicular tubular
atrophy, pancreatic exocrine atrophy
and thymic hyperplasia.
--
Recommended ADI.
0.0011 mg/kg bw/d based on 2 year carcinogenicity in mice (1.1
mg/kg/d with 100- fold UF, three-fold SF for endocrine-related
effects (testicular atrophy, pancreatic
exocrine atrophy), and three-fold for lack of DNT). MOS
for other critical endpoint(s) White matter vacuolation/Neurotox
NOEL for white matter vacuolation was 10 mg/kg/d in chronic dog
study (equivalent to 0.02 mg/kg/d Impurity-5). MOS = 9100. Tumours
NOEL for tumours was 3.8 mg/kg bw/d in 2-year rat study. MOS =
3450. Developmental effects NOAEL for developmental effects was
7 mg/kg bw/d in developmental rabbit study. MOS = 6350
[ MOS = Margin of Safety
]
Ref:
Canada: Regulatory
Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October 27,
2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
Combined chronic toxicity/carcinogenicity
rats. NOAEL = Males: 0.38 mg/kg/day; Females: 0.47 mg/kg/day LOAEL
= Males: 3.8 mg/kg/day; Females: 4.9 mg/ kg/day based on
liver toxicity in both sexes, pancreatic
exocrine atrophy in females and testicular
atrophy in males. Some
evidence of carcinogenicity (thyroid gland follicular cell tumors)
in male rats, but not in females.
Ref: Federal Register: April 18, 2002. Fluazinam;
Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.Apr.18.2002.htm
Chronic toxicity. Fluazinam
was not carcinogenic in rats. A NOAEL of 10 ppm (0.43 mg/kg/day)
of fluazinam was established based on the following effects at
1,000 and/or 100 ppm: lower food consumption and efficiency of
food utilization, slight anemia, elevated cholesterol, increased
liver weights, an increased number of macroscopic liver and testes
lesions and an increased incidence of microscopically observed
lung, liver, pancreas, lymph node
and testes lesions.
Ref: Federal Register: December 6, 2000
[Page 76253-76258]. Notice of Filing Pesticide Petitions to Establish
and to Extend Tolerances for Certain Pesticide Chemicals in or
on Food.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.December.2000.htm
Endocrine:
Testicular (click
on for all fluorinated pesticides)
-- In subchronic and
chronic toxicity studies, fluazinam targeted the following organs:
liver, lung, uterus, testes, pancreas,
thymus, thyroid, stomach, eyes and brain. Generalized toxicity
was observed in rats, mice and dogs as decreases in body weight,
body-weight gain, food consumption and/or food efficiency. Liver
toxicity was evident in most studies including increased size
and weight, fatty changes, pallor, as well as hepatocyte hypertrophy,
necrosis and apoptosis. Thyroid toxicity was less common, but
included follicular hyperplasia and cystic thyroid follicles.
Endocrine-related effects included small and/or flaccid testes,
testicular tubular atrophy,
pancreatic exocrine atrophy and thymic hyperplasia.
--
Recommended ADI.
0.0011 mg/kg bw/d based on 2 year carcinogenicity in mice (1.1
mg/kg/d with 100- fold UF, three-fold SF for endocrine-related
effects (testicular atrophy, pancreatic
exocrine atrophy), and three-fold for lack of DNT). MOS for other
critical endpoint(s) White matter vacuolation/Neurotox NOEL for
white matter vacuolation was 10 mg/kg/d in chronic dog study (equivalent
to 0.02 mg/kg/d Impurity-5). MOS = 9100. Tumours NOEL for tumours
was 3.8 mg/kg bw/d in 2-year rat study. MOS = 3450. Developmental
effects NOAEL for developmental effects was 7 mg/kg bw/d in developmental
rabbit study. MOS = 6350
[ MOS = Margin of Safety
]
Ref:
Canada: Regulatory
Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October 27,
2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
Chronic toxicity. Fluazinam
was not carcinogenic in rats. A NOAEL of 10 ppm (0.43 mg/kg/day)
of fluazinam was established based on the following effects at
1,000 and/or 100 ppm: lower food consumption and efficiency of
food utilization, slight anemia, elevated cholesterol, increased
liver weights, an increased number of macroscopic liver and testes
lesions and an increased incidence of microscopically observed
lung, liver, pancreas, lymph node and testes
lesions. An additional study was conducted to further define the
NOAEL for long-term effects in the rat. In the second study, a
NOAEL of 50 ppm (2.2 mg/kg/day) was established based on liver
and testes effects.
Ref: Federal Register. December 6, 2000.
[PF-983; FRL-6573-7]
http://www.fluoridealert.org/pesticides/Fluazinam.FR.December.2000.htm
-- Combined chronic
toxicity/carcinogenicity rats. NOAEL = Males: 0.38 mg/kg/day;
Females: 0.47 mg/kg/day LOAEL = Males: 3.8 mg/kg/day; Females:
4.9 mg/ kg/day based on liver toxicity in
both sexes, pancreatic exocrine atrophy in females and
testicular atrophy in males.
Some
evidence of carcinogenicity (thyroid gland follicular cell tumors)
in male rats, but not in females.
-- 7-Day inhalation toxicity rats. Test Material: Frowncide WP
(51.9% a.i.). NOAEL = Males: 1.38 mg/kg/day; Females: 1.49 mg/
kg/day LOAEL = Males: 3.97 mg/kg/day; Females: 4.25 mg/ kg/day
based on increased testes weight (males)
and increased liver weight (females).
Ref: Federal Register: April 18, 2002. Fluazinam;
Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.Apr.18.2002.htm
Endocrine:
Thymus (click
on for all fluorinated pesticides)
In
subchronic and chronic toxicity studies, fluazinam targeted the
following organs: liver, lung, uterus, testes, pancreas, thymus,
thyroid, stomach, eyes and brain...
Ref:
Canada: Regulatory Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October 27,
2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
Endocrine:
Thyroid (click on
for all fluorinated pesticides)
Suggestive
Evidence of Carcinogenicity to Humans.
An increase in thyroid gland follicular
cell tumors in male rats, and an increased incidence of
hepatocellular tumors observed in the male mice was treatment-related.
Ref: April
26, 2006 . Chemicals Evaluated for Carcinogenic Potential by the
Office of Pesticide Programs. From: Jess Rowland, Chief Science
Information Management Branch Health Effect Division (7509C) Office
of Pesticide Programs, USEPA.
http://www.fluorideaction.org/pesticides/pesticides.cancer.potential.2006.pdf
Suggestive
Evidence of Carcinogenicity to Humans.
An increase in thyroid gland follicular
cell tumors in male rats, and an increased incidence of
hepatocellular tumors observed in the male mice was treatment-related.
Ref: April
26, 2006 . Chemicals Evaluated for Carcinogenic Potential by the
Office of Pesticide Programs. From: Jess Rowland, Chief Science
Information Management Branch Health Effect Division (7509C) Office
of Pesticide Programs, USEPA.
http://www.fluorideaction.org/pesticides/pesticides.cancer.potential.2006.pdf
-- Combined chronic
toxicity/carcinogenicity rats. NOAEL = Males: 0.38 mg/kg/day;
Females: 0.47 mg/kg/day LOAEL = Males: 3.8 mg/kg/day; Females:
4.9 mg/ kg/day based on liver toxicity in
both sexes, pancreatic exocrine atrophy in females and
testicular atrophy in males.
Some
evidence of carcinogenicity (thyroid gland follicular cell tumors)
in male rats,
but not in females.
Ref: Federal Register: April 18, 2002. Fluazinam;
Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.Apr.18.2002.htm
-- In a carcinogenicity
study in rats, an increased incidence of
thyroid gland follicular cell tumors was observed in males.
In this study, there were statistically significant positive trends
for thyroid gland follicular cell adenocarcinomas and combined
follicular cell adenomas/adenocarcinomas. There was also a statistically
significant increase by pair-wise comparison of the high dose
group (40 mg/kg/day) with the control group for combined follicular
cell adenomas/adenocarcinomas. There was no treatment-related
increase in tumor incidence in female rats. The highest dose level
tested in this study was considered to be adequate but not excessive.
It was concluded that there was some evidence that the thyroid
tumors observed in the male rats in this study were treatment-related.
There were insufficient data to determine whether the thyroid
gland tumors may have been due to disruption
of thyroid-pituitary homeostasis.
Ref: US EPA Pesticide Fact Sheet. Fluazinam.
August 10, 2001.
http://www.epa.gov/opprd001/factsheets/fluazinam.pdf
-- Cancer (oral, dermal,
inhalation). Increases in thyroid
gland follicular cell tumors in male rats; increases in hepatocellular
(liver) tumors in male mice.\2\
-- iii. Cancer. Since fluazinam has been
classified as Suggestive evidence of carcinogenicity, but not
sufficient to assess human carcinogenic potential, an
exposure assessment was not performed.
Ref: Federal Register. September 7, 2001.
Fluazinam; Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.Sept.7.2001.htm
-- In subchronic and
chronic toxicity studies, fluazinam targeted the following organs:
liver, lung, uterus, testes, pancreas, thymus, thyroid,
stomach, eyes and brain... Thyroid toxicity was less common, but
included follicular hyperplasia and cystic
thyroid follicles. Endocrine-related effects included small
and/or flaccid testes, testicular tubular atrophy, pancreatic
exocrine atrophy and thymic hyperplasia.
-- Long-term studies
in both rats and mice provided some evidence of treatment-induced
oncogenicity of the thyroid (follicular
cell adenomas and adenocarcinomas) and liver (hepatocellular
adenomas and carcinomas). Although tumours of this type may be
the result of a non-genotoxic and non-linear mode of action, no
data was provided on potential modes of action (e.g., enzyme induction,
thyroid hormone levels). There was insufficient mechanistic
data to determine the mode of action. A Q was generated at 5.40
¥ 10 -2 in the absence of a demonstrated mode of action.
Ref:
Canada: Regulatory
Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October 27,
2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
Endocrine:
Uterus (click on for
all fluorinated pesticides)
-- In subchronic and
chronic toxicity studies, fluazinam targeted the following organs:
liver, lung, uterus, testes, pancreas,
thymus, thyroid, stomach, eyes and brain...
Ref:
Canada: Regulatory
Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October 27,
2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
-- 90-Day oral toxicity
rats NOAEL: Males = 3.8 mg/kg/day; Females = 4.3 mg/kg/day [[Page
46731]] LOAEL Males = 38 mg/kg/day; Females = 44 mg/kg/day based
on increased liver weights and liver histopathology
in males, and increased lung and uterus
weights in females.
Ref: Federal Register. September 7, 2001.
Fluazinam; Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.Sept.7.2001.htm
Eye
(click on for all fluorinated pesticides)
In subchronic and chronic
toxicity studies, fluazinam targeted the following organs: liver,
lung, uterus, testes, pancreas, thymus, thyroid, stomach, eyes
and brain...
Ref:
Canada: Regulatory
Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October 27,
2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
Acute
eye irritation
rabbits. Technical grade fluazinam. Extremely irritating. Corneal
opacity did NOT reverse in 21 days. Toxicity
Category I.
Ref: US EPA Pesticide Fact Sheet. Fluazinam.
August 10, 2001.
http://www.epa.gov/opprd001/factsheets/fluazinam.pdf
Liver
(click on for all fluorinated pesticides)
Suggestive
Evidence of Carcinogenicity to Humans.
An increase in thyroid gland follicular
cell tumors in male rats, and an increased incidence of hepatocellular
tumors observed in the male mice was treatment-related.
Ref: April
26, 2006 . Chemicals Evaluated for Carcinogenic Potential by the
Office of Pesticide Programs. From: Jess Rowland, Chief Science
Information Management Branch Health Effect Division (7509C) Office
of Pesticide Programs, USEPA.
http://www.fluorideaction.org/pesticides/pesticides.cancer.potential.2006.pdf
In subchronic and chronic
toxicity studies, fluazinam targeted the following organs:
liver, lung, uterus, testes, pancreas, thymus, thyroid,
stomach, eyes and brain... Liver toxicity
was evident in most studies including increased size and weight,
fatty changes, pallor, as well as hepatocyte hypertrophy, necrosis
and apoptosis...
Ref:
Canada: Regulatory
Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October 27,
2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
-- Carcinogenicity
mice NOAEL = Males:1.1 mg/kg/day; Females: 1.2 mg/kg/day [[Page
46732]] LOAEL = Males: 10.7 mg/kg/day; Females: 11.7 mg/kg/day
based on increased incidences of brown macrophages in the liver
of both sexes, eosinophilic vacuolated hepatocytes in males, and
increased liver weight in females.
Clear evidence of carcinogenicity (hepatocellular tumors) in male
mice, but not in females.
-- 90-Day oral toxicity rats NOAEL: Males = 3.8 mg/kg/day; Females
= 4.3 mg/kg/day [[Page 46731]] LOAEL Males = 38 mg/kg/day; Females
= 44 mg/kg/day based on increased liver
weights and liver histopathology
in males, and increased lung and uterus weights in females.
-- 90-Day oral toxicity dogs NOAEL = 10 mg/kg/day LOAEL = 100
mg/kg/day based on retinal effects, increased relative liver
weight, liver histopathology and
possible increased serum alkaline phosphatase in females and possible
marginal vacuolation of the cerebral white matter (equivocal).
-- Prenatal developmental toxicity Maternal NOAEL = 4 mg/kg/day
rabbits LOAEL = 7 mg/kg/day based on decreased food consumption
and increased liver histopathology...
-- Reproduction and fertility effects Parental/Systemic NOAEL
= 1.9 mg/ rats kg/day LOAEL = 9.7 mg/kg/day based on liver
pathology in F1 males...
-- Combined chronictoxicity/ NOAEL = Males: 0.38 mg/kg/day; carcinogenicity
rats Females: 0.47 mg/kg/day LOAEL = Males: 3.8 mg/kg/day; Females:
4.9 mg/kg/day based on liver toxicity
in both sexes, pancreatic exocrine atrophy in females and testicular
atrophy in males. Some evidence of carcinogenicity (thyroid gland
follicular cell tumors) in male rats, but not in females.
-- Carcinogenicity mice NOAEL = Males:<126 mg/kg/day, Females:
<162 mg/kg/day LOAEL = Males: 126 mg/kg/day; Females: 162 mg/kg/day
based on increased liver weights
and liver and brain histopathology
in both sexes Equivocal/some evidence of carcinogenicity (hepatocellular
tumors) in male mice, but not in females,
-- Special studies: 4-Week dietary (Range-finding) rats NOAEL
= Males: 5.1 mg/kg/day; Females: 5.3 mg/kg/day LOAEL = Males:
26.4 mg/kg/day; Females: 25.9 mg/kg/day based on decreased body
weight gain and food consumption, increased serum phospholipids,
increased total cholesterol, increased relative liver
weights, and liver histopathology.
-- 4-Week dietary (Range-finding) mice NOAEL = not identified
(Males; <555 mg/kg/day; Females: <658 mg/ kg/day) LOAEL = Males:
555 mg/kg/day; Females: 658 mg/kg/day based on vacuolation of
white matter in brain, increased liver
weights, histopathology in liver.
-- 90-Day dietary (Special liver
study) rats NOAEL = not determined (Males: <37.6 mg/kg/day, Females:
<44.7 mg/kg/day) LOAEL = Males: 37.6 mg/kg/day, Females: 44.7
mg/kg/day based on increased relative liver
weights and liver histopathology...
Ref: Federal Register. September 7, 2001.
Fluazinam; Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.Sept.7.2001.htm
-- Metabolism and pharmacokinetics
rats. Only 33-40% of the administered dose was absorbed. Most
of the administered dose was recovered in the feces (>89%). Excretion
via the urine was minor (<4%). Total biliary radioactivity, however,
represented 25- 34% of the administered dose, indicating
considerable enterohepatic circulation [Recycling
of certain compounds between the small intestine and the liver].
-- Cancer (oral, dermal, inhalation). `Suggestive evidence of
carcinogenicity, but not sufficient to assess human carcinogenic
potential'' 2 . Quantification of human cancer risk not required.
2 Increases in thyroid gland follicular
cell tumors in male rats; increases in hepatocellular (liver)
tumors in male mice.2 (Ref 2: 2Cancer Assessment Document
- Evaluation of the Carcinogenic Potential of Fluazinam, March
29, 2001, HED Doc. No. 014512.)
-- Cancer. Since fluazinam has been classified as ``Suggestive
evidence of carcinogenicity, but not sufficient to assess human
carcinogenic potential,'' an exposure assessment was not performed.
Ref: Federal Register: April 18, 2002. Fluazinam;
Pesticide Tolerance. Final Rule.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.Apr.18.2002.htm
Lung
(click on for all fluorinated pesticides)
In subchronic and chronic
toxicity studies, fluazinam targeted the following organs: liver,
lung, uterus, testes, pancreas, thymus,
thyroid, stomach, eyes and brain...
Ref:
Canada: Regulatory
Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October 27,
2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
PubMed Abstract: We
report two cases of occupational asthma
caused by sensitisation to powdered fungicides
fluazinam and chlorothalonil,
from the same fungicide formulation plant. Both developed work
related lower respiratory symptoms
after a latent interval of asymptomatic exposure. The diagnosis
in each case was confirmed with a serial peak flow record in the
workplace followed by specific inhalation tests. These fungicides
are known to cause dermatitis; this report indicates that these
compounds can induce specific immunological
reactions in the airways as well as skin.
Ref:
Occup Environ Med 2003 Jan;60(1):76-7. Occupational
asthma from fungicides fluazinam and chlorothalonil; by Draper
A, Cullinan P, Campbell C, Jones M, Newman Taylor A.
-- Subchronic
toxicity. The NOAEL for the 13 week feeding study in rats was
50 ppm (4.1 mg/kg/day). The LOAEL was 500 ppm (41 mg/kg/day),
based on periacinar hepatocellular hypertrophy and sinusoidal
chronic inflammation in males, increased liver weights in males
and increased lung weights in females.
-- Chronic toxicity. Fluazinam was not carcinogenic in rats. A
NOAEL of 10 ppm (0.43 mg/kg/day) of fluazinam was established
based on the following effects at 1,000 and/or 100 ppm: lower
food consumption and efficiency of food utilization, slight anemia,
elevated cholesterol, increased liver weights, an increased number
of macroscopic liver and testes lesions and an increased incidence
of microscopically observed lung,
liver, pancreas, lymph node and testes lesions.
Ref: Federal Register: December 6, 2000
[Page 76253-76258]. Notice of Filing Pesticide Petitions to Establish
and to Extend Tolerances for Certain Pesticide Chemicals in or
on Food.
http://www.fluoridealert.org/pesticides/Fluazinam.FR.December.2000.htm
Spinal
Cord (click
on for all fluorinated pesticides)
Oral 1 year 95.3%,
Beagles, 0, 1, 10, 50 mg/kg/d, 6/sex/dose NOAEL 1 mg/kg/d LOAEL
10 mg/kg/d 10 mg/kg/d 8 Nasal dryness &. 8 Incidence and severity
of gastric lymphoid hyperplasia. 9 Myeloid to erythroid ratios
in bone &. 50 mg/kg/d 8 Nasal dryness and salivation % and &.
9 bwg in both sexes, but only significant in &. 9 Myeloid to erythroid
ratios in bone &. 9 Hematocrit, hemoglobin, RBC % and . 8 WBC
mid and high doses % and &. 8 Alk phos, cholesterol % and &. 8
Abs and rel liver weight % and 8 Incidence
and severity
of white matter vacuolation in brain and spinal cord
8 Incidence and severity of gastric lymphoid hyperplasia.
Ref:
Canada: Regulatory
Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October 27,
2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
Stomach
(click
on for all fluorinated pesticides)
In subchronic and chronic
toxicity studies, fluazinam targeted the following organs: liver,
lung, uterus, testes, pancreas, thymus, thyroid, stomach,
eyes and brain...
Ref:
Canada: Regulatory
Note REG2003-12. Fluazinam. Pest
Management Regulatory Agency. Health Canada. Ottawa. October 27,
2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
|
Environmental
(click on for all
fluorinated pesticides)
--
Ecological Effects Summary:
a. Aquatic (Acute/Chronic Hazard Summary)
Fluazinam is considered to be very
highly toxic to highly toxic to fish (freshwater and estuarine/marine)
on an acute basis (LC50 = 0.036 - 0.11 ppm). Chronic
freshwater NOAEC/LOAEC values were calculated at 0.0053
- 0.00069 ppm and 0.010 - 0.014 ppm, respectively, with
larval survival, reduced number of spawns, and growth as
the endpoints affected. Acute toxicity values for aquatic
invertebrates suggest that fluazinam is highly
toxic to freshwater invertebrates (Daphnia EC50 =
0.18 - 0.22 ppm) and very highly toxic
to estuarine/marine invertebrates (oyster EC50 =
0.0047 and mysid shrimp EC50 = 0.039 ppm ). Chronic toxicity
to invertebrates are only represented through the Daphnia
magna life cycle where the NOAEC was calculated at 0.068
ppm and the LOAEC at 0.140 ppm.. The
endpoints affected for this study were reproductive (reduced
number of young per female) and growth effects.
No acceptable data have been submitted to assess the chronic
effects of fluazinam to estuarine/marine fish or invertebrates.
An estuarine/marine fish life-stage toxicity test (Guideline
72-4a) and an estuarine/marine invertebrate life-cycle toxicity
test (Guideline 72-4b) are required to fulfill these requirements.
-- b. Risk to Aquatic Organisms (Acute/Chronic)
The risk assessment suggests that
exposure of this compound to fish (freshwater and estuarine/marine)
through the proposed use patterns (peanuts and potatoes)
can result in acute (restricted use and endangered species
concern category) and chronic risk. Exposure to aquatic
invertebrates (freshwater and estuarine/marine) from peanut
use can result in acute risk (restricted use and endangered
species concern category). No acute or chronic exceedences
are expected for freshwater invertebrates from the potato
use. Chronic exposure to estuarine/marine fish and invertebrates
could not be calculated at this time because of a lack of
appropriate data.
-- d. Risk to Avian Species (Acute/Chronic)
Although acute exposure should result in minimal toxic effects
to birds, the risk assessment suggests
that the proposed uses can cause chronic (reduced growth
in young) effects in birds. RQ values were calculated
for exposure to peanuts (maximum EECs RQ = 1.0 - 1.8 and
56 day average EECs RQ = 1.1 ppm) and potatoes (maximum
EECs RQ = 1.0 - 1.5 and 56 day average(RQ = 1).
-- e. Risk to Mammalians (Acute, Chronic)
The risk assessment suggests that the proposed uses can
result in chronic risk to mammalians
(herbivores and insectivores). RQ values were calculated
for exposure to peanuts (maximum EECs RQ = 1.6 - 3.5 and
56 day average EECs RQ = 1.0 - 2.2) and potatoes (maximum
EECs RQ = 1.0 - 1.9 and 56 day average EECs RQ = 1.4 - 3.0).
Acute concerns appear to be focused on grass eating endangered
mammals (RQ = 0.1)
Ref: US EPA Pesticide Fact
Sheet. Fluazinam. August 10, 2001.
http://www.epa.gov/opprd001/factsheets/fluazinam.pdf
--
Risk mitigation. Fluazinam is of very
high toxic risk to freshwater aquatic organisms, of high
toxic risk to marine aquatic organisms, and of high dietary
and reproductive risk to wild mammals.
-- Results of terrestrial field studies of dissipation
and accumulation conducted in Canada indicated that fluazinam
was moderately persistent in soil,
with DT50 values of 81.5 and 95 days with significant carryover
of residues to the next season. The major transformation
product, HYPA, was formed at one of the Canadian field sites
reaching a maximum of 15%. HYPA was a minor transformation
product at the other Canadian site. There was no evidence
of leaching of fluazinam or HYPA through the soil layers.
Field dissipation studies conducted in the U.S. yielded
DT50 values of 19 and 33, indicating slight persistence.
HYPA was formed as a major transformation product at one
of the U.S. sites reaching 15% HYPA was a minor transformation
product at the other U.S. site. Both field and laboratory
studies indicate that fluazinam will be moderately persistent
and have low potential to leach.
Ref:
Canada:
Regulatory Note REG2003-12. Fluazinam.
Pest Management Regulatory Agency.
Health Canada. Ottawa. October 27, 2003.
http://www.fluorideaction.org/pesticides/fluazinam.canada.report2003.pdf
|
A
February
16, 2005,
check at the Code
of Federal Regulations for Fluazinam (and its metabolite
in or on grape): this fungicide is permitted in
or on 3 food
commodities in the United States.
The
following list identifies these crops for which EPA has set
pesticide tolerances.
Note:
On April 18, 2002, US EPA established an import
tolerance for residues of fluazinam and its metabolite
AMGT3 in or on wine grapes at 3.0 ppm. ISK BioSciences Corporation
requested this tolerance under the Federal Food, Drug, and
Cosmetic Act, as amended by the Food Quality Protection
Act of 1996.
|
| [Code
of Federal Regulations]
[Title 40, Volume 22]
[Revised as of July 1, 2004]
From the U.S. Government Printing Office via GPO Access
[CITE: 40CFR180.574]
[Page 510-511]
TITLE 40--PROTECTION OF ENVIRONMENT
CHAPTER I--ENVIRONMENTAL PROTECTION AGENCY (CONTINUED)
PART 180_TOLERANCES AND EXEMPTIONS FROM TOLERANCES FOR PESTICIDE
CHEMICALS
IN FOOD--Table of Contents
Subpart C_Specific Tolerances
Sec. 180.574 Fluazinam; tolerances for residues.
(a)(1) General. Tolerances are established for residues of
fluazinam, (3-
chloro-N-[3-chloro-2,6-dinitro-4-(trifluoromethyl) phenyl]-5-
(trifluoromethyl)-2-pyridinamine) in or on the following commodities: |
|
Commodity |
Parts
per million |
Peanut
|
0.02 |
| Potato |
0.02 |
| (a)(2)
Tolerances are established for residues
of fluazinam and its
metabolite AMGT 3-[[4-amino-3-[[3-chloro-5-(trifloromethyl)-2-
pyridinyl]amino]-2-nitro-6-(trifluoromethyl) phenyl] thio]-2-(beta-D-
glucopyranosyloxy) propionic acid) in or on the following
commodity: |
| Wine grape
/1/ |
3.0 |
| /1/
No US registration as of March 15, 2002. |
| (b)
Section 18 emergency exemptions. [Reserved]
(c) Tolerances with regional registrations. [Reserved]
(d) Indirect or inadvertent residues. [Reserved] |
|

