September 19, 2004
Philadelphia Inquirer
Teflon mystery
raises safety questions
The acid is
in the environment and in children's blood, but scientists don't know how
it got there. The health risks are unknown.
By Tom Avril
Inquirer Staff Writer
More than 65 years ago
in a South Jersey laboratory, a DuPont Co. chemist accidentally invented
a waxy, white powder that would become one of the mainstays of the modern
kitchen: Teflon.
Today, this nonstick
marvel is getting attention far beyond the stove-top.
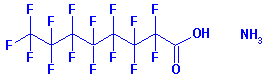
A chemical used to make
it, perfluorooctanoic acid (PFOA), has been turning up in people and animals
worldwide: river otters in Oregon, polar bears in the Canadian Arctic, and
in the blood of 96 percent of children tested in 23 states.
Scientists are not sure
how the chemical is getting into people - not from using Teflon pans, they
say - and they don't know whether it poses any danger at current levels.
But the hardy substance
does not break down in the environment. And it has been linked to liver
and developmental problems in lab rats, prompting an unusually broad review
by the U.S. Environmental Protection Agency.
"We need to get
to the bottom of this," said Charles Auer, director of the agency's
Office of Pollution Prevention and Toxics.
PFOA is just one of
dozens of synthetic intruders found in the human body in recent years, thanks
to increasingly sophisticated equipment that allows scientists to measure
the merest traces of chemicals.
But this one has drawn
unusual scrutiny.
DuPont agreed this month
to pay at least $108 million to settle a class-action suit brought by residents
near a company plant in West Virginia, where PFOA has been found in the
drinking water.
And in July, the EPA
- prompted partly by the research of a Washington-based nonprofit called
Environmental Working Group - accused DuPont of failing to disclose certain
health-related information about PFOA from as early as 1981. A public hearing
is pending, after which an administrative law judge can impose fines of
more than $300 million.
Meanwhile, a growing
number of scientists, among them a team at the University of Toronto, think
the biggest source of PFOA is not Teflon manufacture, but a related family
of chemicals called telomers.
Telomers, made by DuPont
and a handful of other companies, are widely used to make grease- and stain-repellent
coatings for take-out food boxes, carpets, and clothing.
Burger
King, for example,
stopped selling food in telomer-coated boxes in 2002. McDonald's
has said it uses such boxes, but would not say whether it still does.
The various accusations
have provoked consternation at DuPont, based in Wilmington, whose executives
describe it as a "science company" that cares about the environment.
The company says it
has broken no laws and has sharply reduced emissions of PFOA. And studies
on plant workers have shown PFOA to be safe, said Don Duncan, president
of the Society of the Plastics Industry, an industry group.
"It's not as if
we've got people dropping in the streets out there," he said.
•
In April 1938, Roy Plunkett was experimenting with refrigerants in a DuPont
lab in Deepwater, N.J., when something strange happened.
His assistant opened
a pressurized metal cylinder to release the gas stored inside, but nothing
came out.
The cylinder was too
heavy to be empty. Intrigued, they turned it upside down. A mysterious white
powder fell out.
The gas molecules somehow
had combined to form much longer polymer chains - a new plastic that would
turn out to be more slippery than ice.
Company scientists eventually
developed a way to repeat the accident in mass quantities, with a process
using another chemical, PFOA. Eventually, Teflon and related substances
were used to make pots and pans, medical equipment, airplane parts, and
automobile fuel lines.
Then, in 1968, 30 years
after Plunkett's discovery, a University of Rochester scientist noticed
something odd.
Don Taves, a toxicologist,
was testing human blood to see how much fluorine was in it as a result of
fluoridated water, which prevents cavities.
The numbers were 10
times what he expected, recalled Taves, now 78. After further research,
he became convinced that synthetic perfluorochemicals were at fault.
But Taves said he was
not concerned about any health effects, and the findings attracted little
attention until 1997.
That year, 3M Co., the
Minnesota chemical-maker that made PFOA and sold it to DuPont, was measuring
its workers' blood for a related chemical that was produced during the manufacture
of Scotchgard.
Company scientists obtained
samples from nonworkers as a baseline to compare the workers' blood. To
their surprise, the chemical was present in nonworkers, too.
In 2000, the company
said it would voluntarily stop making the chemical, perfluorooctane sulfonate,
and find a new way to make Scotchgard. At the same time, 3M said it would
stop making a related chemical that scientists had been finding in the environment:
PFOA.
Faced with the loss
of its key supplier, DuPont decided to start making PFOA itself.
Today, the DuPont plant
in Fayetteville, N.C., is the nation's lone manufacturer of PFOA.
•
Sitting in a freezer in a University of Pennsylvania laboratory, hundreds
of vials of blood may hold part of the answer to the big question:
Is PFOA a risk to human
health?
A team led by Edward
A. Emmett, a professor of occupational and environmental medicine, is collecting
the blood samples from people who live near the DuPont plant in Parkersburg,
W.Va.
With a government grant,
the team will analyze the samples to see whether there is a connection between
PFOA and hormone levels and liver function.
Though various industry
studies have found no health effects from PFOA, Emmett said some of the
studies were small or not well-designed.
"We're at a pretty
rudimentary stage in our knowledge," he said.
And past studies on
lab animals have drawn an incomplete picture, in part because their bodies
do not handle the chemical in the same way.
For
example, the half-life of PFOA in humans is believed to be about four years,
meaning it takes that long for people to flush half of the chemical out
of their systems. Rats get rid of the chemical in a matter of days,
leaving the interspecies comparisons open to a wide range of educated guesswork.
In a preliminary assessment
last year of risks to human development, the EPA found the chemical levels
linked to problems in rats to be anywhere from less
than 70 to more than 9,000 times the levels found in women and children.
For some subtle health
effects, including a weaker immune system and low organ weights, scientists
have found no dose of the chemical that is totally safe in rats.
Rather than wait for
more studies, critics advocate a precautionary approach: Stop making the
suspect chemicals.
"We're already
to the point where it is in people and getting near the point where there's
significant risk," said Tim Kropp, a toxicologist at Environmental
Working Group.
Meanwhile, industry,
university and government scientists are hard at work trying to solve the
other mystery: How do PFOA and other perfluorochemicals get into people
to begin with?
Could it be from the
water? The air? Dust from vacuuming stain-resistant carpets? Suddenly, it's
a hot research topic.
In the last three years,
journals publishing papers on perfluorochemicals have grown tenfold, to
nearly 50, according to the journal Environmental Science & Technology.
"Scientists are
way behind," said Keri Hornbuckle, a University of Iowa engineering
professor. "We're scurrying to figure out all the chemical pathways
that these chemicals go through."
One possibility: When
people wash their stain- or water-resistant clothes,
the chemical coatings may go down the drain and into the environment.
Another theory is that
some residues escape into the air, where they break
down into PFOA or similar substances - some of them thought to pose
even greater risk.
In a study awaiting
publication, DuPont scientists say they find no risk associated with the
everyday use of coated clothing, carpets and cookware, among other products.
PFOA is not present in these final products except sometimes in trace amounts.
"We can say unequivocally
that those articles are safe," said Robert C. Buck, a Ph.D. chemist
and senior research scientist for the company.
And the FDA, which has
approved 30 perfluorochemicals for food packaging and processing since 1958,
says there is no sign of a direct risk from food.
But exposure from package
disposal or deterioration must still be ruled out, said George Pauli, a
senior official at the FDA's Office of Food Additive Safety.
In short, things have
gotten much more complicated since the birth of Teflon in 1938.
The perfluorochemicals
are heralded for their durability and nonstick qualities, but some of them
may be sticking around too long.
------------------------------------------------------------------------
Contact staff writer
Tom Avril at 215-854-2430 or tavril@phillynews.com.