|
Return
to Gamma
cyhalothrin Index Page
Activity:
Insecticide (pyrethroid)
Structure:
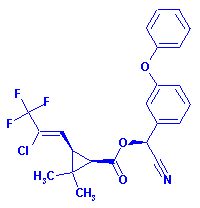
Adverse
Effects:
Body Weight Decrease
Dermal
Endocrine:
Suspected Disruptor
Neuromuscular
Reproductive
US:
Lambda-cyhalothrin and its isomer Gamma-cyhalothrin have
more than 380 food tolerances, including:
Alfalfa, Egg, Garlic,
Barley, Brassica, Cattle, Corn, Cotton, Goat, Hog, Horse,
Lettuce, Milk, Onion, Peanut, Poultry, Rice, Sheep, Sorghum,
Soybean, Sugarcane, Sunflower Seeds, Tomato, Wheat.
See
tolerances at: http://www.fluorideaction.org/pesticides/mrls.lambda-cyhalothrin.htm
*
October 2003: very little toxicological data accessible.
However, a number of studies have been performed and submitted
to US EPA. See list below.
-- On
April 8, 2004, the Federal Register published a Final Rule
for the first-time pesticide tolerances
for the isomer form of gamma-cyhalothrin. Gamma-cyhalothrin
is the isolated active isomer of lambda-cyhalothrin. Pytech
Chemicals GmbH, 9330 Zionsville Rd., Indianapolis,
IN 46268, requested this change in tolerance expression
in support of the registration of a pesticide formulation
enriched with the gamma isomer of lambda-cyhalothrin.
Gamma-cyhalothrin
is a single, resolved isomer of the pyrethroid insecticide
cyhalothrin, and as such shares physical, chemical, and
biological properties with both cyhalothrin and lambda-cyhalothrin,
which are mixtures of 4 and 2 isomers respectively. Gamma-cyhalothrin
is the most insecticidally active isomer of cyhalothrin/lambda-cyhalothrin,
and thus the technical gamma-cyhalothrin product may be
considered a refined form of cyhalothrin/lambda-cyhalothrin
in that it has been purified by removal of less active and
inactive isomers. Thus,
similar levels of insecticidal efficacy for gamma-cyhalothrin
can be obtained with significantly reduced application rates
as compared with either cyhalothrin or lambda-cyhalothrin.
US
former tolerances for identifies lambda-cyhalothrin as a
1:1 mixture of two isomers and their epimers, one of which
is the gamma isomer. The gamma isomer is present at 42%
in this mixture. By contrast in the proposed tolerance expression
the gamma isomer is present at 98% in the mixture.
-- It is noted that
in the developmental toxicity study in the rat that the
resolved gamma
isomer is over an order of magnitude more toxic than in
cyhalothrin.
-- International Residue Limits
There are currently no Mexican, Canadian, or Codex MRLs
(maximum residue limits) for gamma- or lambda-cyhalothrin;
however, there are MRLs for cyhalothrin
from which lambda-cyhalothrin is derived as an enriched
isomer. A Codex MRLs of 0.2 part per million (ppm) has been
established
for pome fruits for cyhalothrin, which is inconsistent with
the proposed U.S. lambda-cyhalothrin tolerance of 0.3 ppm
for pome fruits. It is unclear if harmonization can be achieved
because residues up to 0.25 ppm were found in the U.S. trials
for apples. Codex MRLs were not established
for the other crops presently under consideration.
Ref: US EPA. Lambda-Cyhalothrin
and an Isomer Gamma-Cyhalothrin; Tolerances for Residue.
FINAL RULE. Federal
Register.
|
Body
Weight Decrease (click
on for all fluorinated pesticide)
Reproductive
and developmental toxicity.
A developmental toxicity study in rats given gavage doses of 0,
0.1, 0.5, and 2 mg/kg/day with no developmental toxicity
observed under the conditions of the study. The developmental
no observed adverse effect level (NOAEL) is greater than 2
mg/kg/day, the highest dose tested (HDT).
The maternal NOAEL and lowest observed adverse
effect level (LOAEL) are established at 0.5 and 2 mg/kg/day, respectively,
based on reduced body weight, body
weight gain, and feed consumption.
Subchronic
toxicity.
A 90-day feeding study in rats fed doses of 0, 2.5, 10, 50, and
100 parts per million (ppm) with a
NOAEL of 50 ppm and a LOAEL of 100 ppm based
on mortality, decreased feed consumption, decreased body weights,
and increased relative liver and kidney
weight at 100 ppm.
Ref:
Federal Register: February 25, 2004. Gamma-Cyhalothrin; Notice
of Filing a Pesticide Petition to Establish a Tolerance for a
Certain Pesticide Chemical in or on Food
http://www.fluorideaction.org/pesticides/gamma.cyhal.fr.feb25.2004.htm
Dermal
(click
on for all fluorinated pesticide)
13-Week Dietary
- Rat.
NOAEL:
male/female =3.4/4.2 mg/kg/day
LOAEL: male/female = 6.6/8.8 mg/kg/day (mortality
in males, neuromuscular effects in both sexes,
dermatitis, and gross and microscopic skin lesions in females).
Ref:
April 8, 2004. Federal Register. Lambda-Cyhalothrin and Gamma-Cyhalothrin.
Tolerances for Residues. FINAL RULE.
http://www.fluorideaction.org/pesticides/cyhalothrin.fr.april.8.2004.htm
Endocrine:
Suspected Disruptor
(click
on for all fluorinated pesticide)
Suspected
Endocrine
Disruptor
Ref:
PAN
Pesticides Database
Neuromuscular
(click
on for all fluorinated pesticide)
13-Week Dietary
- Rat (Gamma cyhalothrin).
NOAEL:
male/female =3.4/4.2 mg/kg/day
LOAEL: male/female = 6.6/8.8 mg/kg/day (mortality
in males, neuromuscular effects in both
sexes, dermatitis, and gross and microscopic skin lesions
in females).
Ref:
April 8, 2004. Federal Register. Lambda-Cyhalothrin and Gamma-Cyhalothrin.
Tolerances for Residues. FINAL RULE.
http://www.fluorideaction.org/pesticides/cyhalothrin.fr.april.8.2004.htm
Reproductive
(click
on for all fluorinated pesticide)
3-Generation
Reproduction - Rat (Cyhalothrin; Lambda cyhalothrin
and Gamma cyhalothrin).
Parental NOAEL: 1.5 mg/kg/day; Parental
LOAEL: 5.0 mg/kg/day
(decreased parental body weight and body weight gain during premating
and gestation periods).
Reproductive NOAEL: 5.0 mg/kg/day; Reproductive LOAEL: Not established.
Offspring NOAEL: 1.5 mg/kg/day
Offspring LOAEL: 1.5 mg/kg/day
(reduced pup weight and weight gain during lactation).
Ref:
April 8, 2004. Federal Register. Lambda-Cyhalothrin and Gamma-Cyhalothrin.
Tolerances for Residues. FINAL RULE.
http://www.fluorideaction.org/pesticides/cyhalothrin.fr.april.8.2004.htm
Request
from FAN:
If you have copies of the following reports, please share
them with us. thanks. EC.
|
INITIAL
SUBMISSION: LETTER FROM [] TO USEPA RE RESULTS OF ACUTE
DIETARY TOXICITY STUDY IN RATS OF CYCLOPROPANECARBOXYLIC
ACID 3-(2-CHLORO-3,3,3-TRIFLUOR*,
DATED 11-02-99 (SANITIZED)
Keywords:
CONFIDENTIAL
CYCLOPROPANECARBOXYLIC ACID, 3-(2-CHLORO-3,3,3-TRIFLUORO-1-*
HEALTH EFFECTS
ACUTE TOXICITY
MAMMALS
RATS
ORAL DIET
Source:
EPA/OTS;
Doc #88000000026S
CAS
No:
76703-62-3
Order
Number: NTIS/OTS0559833
|
INITIAL
SUBMISSION: LETTER FROM [] TO USEPA RE RESULTS IN REPEATED
DOSE (ORAL GAVAGE) DEVELOPMENTAL TOXICITY PROBE IN RATS
W/CYCLOPROPANECARBOXYLIC ACID*, DATED
12-06-99 (SANITIZED)
Keywords:
CONFIDENTIAL
CYCLOPROPANECARBOXYLIC ACID, 3-(2-CHLORO-3,3,3-TRIFLUORO-1-*
HEALTH EFFECTS
REPRODUCTION/FERTILITY EFFECTS
TERATOGENICITY
MAMMALS
RATS
ORAL
GAVAGE
SUBCHRONIC TOXICITY
Source:
EPA/OTS; Doc #88000000047S
CAS
No: 76703-62-3
Order Number: NTIS/OTS0559854
|
INITIAL
SUBMISSION: LETTER FROM [] TO USEPA REPORTING RESULTS OF
EYE IRRITATION STUDY IN A SOLITARY RABBIT WITH CYCLOPROPANECARBOXYLIC
ACID, 3-*, DATED 6/7/00 (SANITIZED)
Keywords:
CONFIDENTIAL
CYCLOPROPANECARBOXYLIC ACID, 3-(2-CHLORO-3,3,3-TRIFLUORO-1-*
HEALTH EFFECTS
PRIMARY EYE IRRITATION
MAMMALS
RABBITS
DERMAL
Source:
EPA/OTS; Doc #88000000180S
CAS
No: 76703-62-3
Order Number: NTIS/OTS0559941
|
INITIAL
SUBMISSION: LETTER FROM [] TO USEPA REPORTING CNS EFFECTS
IN RANGE-FINDING STUDIES OF CYCLOPROPANECARBOXYLIC ACID,
3-(2-CHLORO-3,3,3-TRIFLUORO-*, DATED
03/15/00 (SANITIZED)
Keywords:
CONFIDENTIAL
CYCLOPROPANECARBOXYLIC ACID, 3-(2-CHLORO-3,3,3-TRIFLUORO-1-*
HEALTH EFFECTS
SUBCHRONIC TOXICITY
MAMMALS
MICE
ORAL
NEUROTOXICITY
PRIMARY DERMAL SENSITIZATION
GUINEA PIGS
DERMAL
Source:
EPA/OTS; Doc #88000000122S
CAS No: 76703-62-3
Order Number: NTIS/OTS0574034
|
INITIAL
SUBMISSION: LETTER FROM [] TO USEPA RE RESULTS OF SUBACUTE
DIETARY TOXICITY STUDY OF CYCLOPROPANECARBOXYLIC ACID, *
IN BOBWHITE & MALLARDS, DATED 11/28/2000
(SANITIZED)
Keywords:
CONFIDENTIAL
CYCLOPROPANECARBOXYLIC ACID, 3-(2-CHLORO-3,3,3-TRIFLUORO-1-*
ENVIRONMENTAL EFFECTS
ACUTE TOXICITY
BIRDS
Source:
EPA/OTS; Doc #88010000044S
CAS
Nos: 76703-62-3
Order Number: NTIS/OTS0574112
|
|

