|
Return
to Benfluralin
Index Page
Activity: Herbicide
(2,6-dinitroaniline)
Structure:
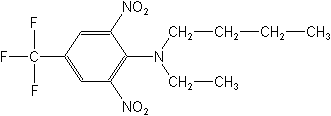
Adverse
Effects:
Ataxia
Blood
Body Weight Decrease
Cancer. Suggestive
Evidence of Carcinogenicity: LIVER.
Endocrine: Thyroid
Kidney
Liver
Spleen
Environmental
As
of February 15, 2005, this herbicide is permitted to have
residues in or on 8 food commodities in the US.
They are presented at the bottom of this page.
|
Ataxia
(click
on for all fluorinated pesticides)
Animal Toxicity Studies:
... ATAXIA, WEAKNESS, FALLING. REGURGITATION
ALSO OCCURRED IN FIRST 2 HR AFTER TREATMENT. THE OTHER SIGNS OCCURRED
2 TO 14 DAYS AFTER TREATMENT. ALTHOUGH NO BIRDS DIED, CONSIDERABLE
WT LOSSES HAD OCCURRED BY THE END OF THE 14 DAY OBSERVATION PERIOD.
/FEMALE MALLARDS, ACUTE ORAL/ [U.S. Department of the Interior,
Fish and Wildlife Service. Handbook of Toxicity of Pesticides
to Wildlife. Resource Publication 153. Washington, DC: U.S. Government
Printing Office, 1984. 13]
Ref: TOXNET profile from Hazardous Substances
Data Bank.
http://www.fluoridealert.org/pesticides/Benfluralin.TOXNET.HSDB.htm
Blood
(click on for all fluorinated pesticides)
Increased relative
liver weights, decreased red blood cell
counts and decreased hematocrit and
hemoglobin levels were observed in dogs orally administered
benfluralin at a dose of 125 mg/kg/day for 2 years. The NOAEL
was 25 mg/kg/day. Based on the NOAEL, EPA has established an oral
RfD of 0.003 mg/kg/day. EPA believes that there is sufficient
evidence for listing benfluralin on EPCRA section 313 pursuant
to EPCRA section 313(d)(2)(B) based on the available hematological
toxicity data for this chemical.
Ref:
USEPA/OPP. Support Document for the Addition of Chemicals from
Federal Insecticide, Fungicide, Rodenticide Act (FIFRA) Active
Ingredients to EPCRA Section 313. U. S. Environmental Protection
Agency, Washington, DC (1993). As cited by US EPA in: Federal
Register: January 12, 1994. Part IV. 40 CFR Part 372. Addition
of Certain Chemicals; Toxic Chemical Release Reporting; Community
Right-to-Know; Proposed Rule.
Body
Weight Decrease
(click
on for all fluorinated pesticides)
--
REPRODUCTION, RAT **208-078 167287, ATwo-Generation Reproduction
Study in Rats with Benefin@, (Janet A. Trutter, Hazleton Washington,
Inc., Vienna, VA., Report # HWA 174-136, 9 February 1995). Thirty
Sprague-Dawley Crl:CD 7 BR rats per sex per group received benefin
(95.8% purity) in the diet at concentrations of 0, 100, 1000,
and 5000 ppm through 2 generations with one litter per generation.
Parental NOEL = 100 ppm (7.1 and 8.8 mg/kg/day for M and F, respectively,
based on hepatocellular enlargement, enhanced extent of chronic
progressive nephropathy in both sexes, and renal tubule hyaline
droplets in males: also associated increased liver and kidney
weights). Offspring NOEL = 100 ppm (reduced
body weights of pups by day 7, continuing at least through weaning).
The high dose caused pup weight reductions
at weaning to about 60% of controls. Adult 5000 ppm F1
males (the more sensitive gender and generation of adults) weighed
about 80% of controls after 10 to 19 weeks of treatment. Lesser
but statistically significant decrements were seen in F0 males
and in F0 and F1 females at 5000 ppm. Food consumption
was correspondingly reduced in both sexes at this dose level.
Offspring effects limited to 5000 ppm included significant
reduction of mean live litter size at birth and a reduction
of pup viability between day 4 cull and weaning (21% loss in 5000
ppm F2 pups). Acceptable, with no adverse effects. (H. Green and
C. Aldous, 4/21/00).
-- TERATOLOGY, RABBIT **208-077 167286, ÒTeratology Study in Rabbits
with BenefinÓ, (M. D. Mercieca, Springborn Laboratories, Inc.,
Spencerville, OH, Report # 3130.9, 3 June 1991). Twenty inseminated
female New Zealand white rabbits per group received benefin (in
10% aqueous acacia) at concentrations of 0, 25, 50, 100, and 225
mg/kg/day on gestation days 6 through 18 in a guideline teratology
study. Maternal NOEL = 50 mg/kg/day (increased incidence of does
with few or no feces). Developmental NOEL = 225 mg/kg/day (highest
dose tested). The highest dose was clearly maternally toxic, as
indicated by significant (p < 0.01) reductions
in food consumption and body weight gain, and by the presence
of 3 aborted litters and one total litter resorption loss. Acceptable,
with no adverse effects (H. Green, and C. Aldous, 4/18/00).
Ref: Summary of Toxicological Data: Benefin.
California EPA, Department of Pesticide Regulation, Medical Toxicology
Branch. Revised April 21, 2000.
http://www.fluorideaction.org/pesticides/benefin.ca.tox.rev.apr.2000.pdf
Animal Toxicity Studies:
... ATAXIA, WEAKNESS, FALLING. REGURGITATION
ALSO OCCURRED IN FIRST 2 HR AFTER TREATMENT. THE OTHER SIGNS OCCURRED
2 TO 14 DAYS AFTER TREATMENT. ALTHOUGH NO BIRDS DIED, CONSIDERABLE
WT LOSSES HAD OCCURRED BY THE END OF THE 14 DAY OBSERVATION
PERIOD. /FEMALE MALLARDS, ACUTE ORAL/ [U.S. Department of the
Interior, Fish and Wildlife Service. Handbook of Toxicity of Pesticides
to Wildlife. Resource Publication 153. Washington, DC: U.S. Government
Printing Office, 1984. 13]
Ref: TOXNET profile from Hazardous Substances
Data Bank.
http://www.fluoridealert.org/pesticides/Benfluralin.TOXNET.HSDB.htm
Cancer;
LIVER (click
on for all fluorinated pesticides)
Suggestive
Evidence of Carcinogenicity, but Not Sufficient to Assess Human
Carcinogenic Potential. Liver tumors (hepatocellular adenomas
& carcinomas) in 2 genetically related strains of mice (CD-1
& Swiss SPF) (M & F).
Ref: April
26, 2006 . Chemicals Evaluated for Carcinogenic Potential by the
Office of Pesticide Programs.
From: Jess Rowland, Chief Science Information Management Branch
Health Effect Division (7509C) Office of Pesticide Programs, USEPA.
http://www.fluorideaction.org/pesticides/pesticides.cancer.potential.2006.pdf
Endocrine:
Thyroid
(click
on for all fluorinated pesticides)
208-079 167288,
ÒBenefin: Two-Year Dietary Chronic Toxicity/Oncogenicity Study
in Fischer 344 RatsÓ, (Michael R. Moore, Corning Hazleton Inc,
Vienna, VA., Laboratory ID # CHV 174-133, Sponsor Report # DR-0097-3397-005,
1 July 1996). Fifty CDF ¨ (F-344)CrlBR rats per sex per group
received benefin in the diet at 0, 10, 100, 2500, and 5000 ppm
for 2 yr. An additional 10 per sex per group at the same dose
levels were designated for 1-yr interim sacrifice. Estimated achieved
dosages were 0, 0.5, 5.4, 136, and 275 mg/kg/day in males, and
0, 0.7, 6.8, 168, and 331 mg/kg/day in females, respectively over
weeks 1-104....Tumor incidence: thyroid
follicular tumors (adenomas and carcinomas) increased in
males (incidences of 1, 1, 1, 7,
and 8 in controls through high dose, respectively) and females
(incidences of 0, 0, 1, 5, and 4)... Tumors are possible adverse
effects, which should be evaluated in perspective of the many
indications of excessive exposures at effective dose levels. Green
and Aldous, 4/11/00.
Ref:
Summary of Toxicological Data: Benefin. California EPA, Department
of Pesticide Regulation, Medical Toxicology Branch. Revised April
21, 2000.
http://www.fluorideaction.org/pesticides/benefin.ca.tox.rev.apr.2000.pdf
Kidney
(click
on for all fluorinated pesticides)
COMBINED, RAT **208-079
167288, ÒBenefin: Two-Year Dietary Chronic Toxicity/Oncogenicity
Study in Fischer 344 RatsÓ, (Michael R. Moore, Corning Hazleton
Inc, Vienna, VA., Laboratory ID # CHV 174-133, Sponsor Report
# DR-0097-3397-005, 1 July 1996). Fifty CDF ¨ (F-344)CrlBR rats
per sex per group received benefin in the diet at 0, 10, 100,
2500, and 5000 ppm for 2 yr. An additional 10 per sex per group
at the same dose levels were designated for 1-yr interim sacrifice.
Estimated achieved dosages were 0, 0.5, 5.4, 136, and 275 mg/kg/day
in males, and 0, 0.7, 6.8, 168, and 331 mg/kg/day in females,
respectively over weeks 1-104. Chronic NOEL = 10 ppm (0.5 and
0.7 mg/kg/day in M and F), based on increased
hyaline droplets in kidneys of both sexes at 100 ppm and above,
increased and tubular cell karyomegaly and transitional cell hyperplasia
in kidneys of males, hepatocellular hypertrophy and increased
hepatocellular pigmentation in females, and calculus of renal
pelvis in females. Substantial toxicity prompted investigators
to determine that the two highest dose levels Òexceeded the maximum
tolerated dose and therefore should not be used for risk assessmentÓ.
The study design, with no dose levels between 100 and 2500 ppm,
gave no opportunity to characterize dose-response in this range
of primary interest. These were major changes at the highest two
dose levels (in both sexes, unless indicated), in addition to
the findings for the NOEL (above) applying to both sexes. Body
weights were reduced over time (in males, primarily in last few
weeks of the study), so that at termination the deficits of 2500
and 5000 ppm groups compared to controls were 8 and 17% in males,
and 18 and 28% in females, respectively. In females only, this
was accompanied by approximately 10% food consumption reductions
at each of the higher dose levels. There were reductions in the
main hematology parameters (RBC counts, HCT, and Hb levels). Incidence
and/or degree of chronic progressive nephropathy
was increased. The majority of these rats had ÒslightÓ to ÒminimalÓ
degeneration of sciatic nerve and skeletal muscle (thigh). Males
had elevated incidence of single cell necrosis in liver.
Chronic inflammation of the lungs was seen in the majority of
these rats, whereas low incidences of congestion of the
abdominal cavity, and urinary bladder hyperplasia were limited
to these dose levels. Numerous clinical chemistry changes mirrored
the above pathology in kidneys, liver,
and perhaps other tissues at the higher two dose levels. Tumor
incidence: thyroid follicular tumors (adenomas and carcinomas)
increased in males (incidences of 1, 1, 1, 7, and 8 in controls
through high dose, respectively) and females (incidences of 0,
0, 1, 5, and 4). Hepatocellular tumors (primarily adenomas) were
increased in males (incidences of 2, 2, 1, 5, and 11 in controls
through high dose, respectively). There were no statistically
significant increases in epithelial cell tumors in kidneys nor
urinary bladder, however the low incidences of transitional cell
papilloma and tubule cell adenoma or carcinoma
were predominantly found in 2500 and 5000 ppm groups (compare
to the congener, trifluralin). Study is acceptable. Tumors are
possible adverse effects, which should be evaluated in perspective
of the many indications of excessive exposures at effective dose
levels. Green and Aldous, 4/11/00.
Ref:
Summary of Toxicological Data: Benefin. California EPA, Department
of Pesticide Regulation, Medical Toxicology Branch. Revised April
21, 2000.
http://www.fluorideaction.org/pesticides/benefin.ca.tox.rev.apr.2000.pdf
Liver
(click
on for all fluorinated pesticides)
"Benefin: 13-Week Oral
Toxicity Study in Beagle Dogs", (D.W. Dalgard, Hazleton Washington,
HWA 174-135, 12/28/93). Benefin, purity 95.8%, administered via
capsule at concentrations of 0, 5, 25, 125 mg/kg/day to 4 Beagle
dogs/sex/group for 13 weeks. Emesis (compound colored) was slightly
increased at all dose levels for females and for mid and high
dose males. Liver weights was increased
for high dose males; and the incidence of
hepatocellular hypertrophy was increased for high dose
males and females. Increased incidence of hemosiderin pigment
was observed in the liver at the
high dose and the spleen at mid and high dose levels. NOAEL =
5 mg/kg/day. Acceptable (Kishiyama, J., and P. Iyer, 5/12/98).
Ref: Summary of Toxicology Data for Benefin.
California EPA, Department of Pesticide Regulation, Medical Toxicology
Branch
http://www.cdpr.ca.gov/docs/toxsums/pdfs/53.pdf
COMBINED, RAT **208-079
167288, "Benefin: Two-Year Dietary Chronic Toxicity/Oncogenicity
Study in Fischer 344 Rats", (Michael R. Moore, Corning Hazleton
Inc, Vienna, VA., Laboratory ID # CHV 174-133, Sponsor Report
# DR-0097-3397-005, 1 July 1996). Fifty CDF ¨ (F-344)CrlBR rats
per sex per group received benefin in the diet at 0, 10, 100,
2500, and 5000 ppm for 2 yr. An additional 10 per sex per group
at the same dose levels were designated for 1-yr interim sacrifice.
Estimated achieved dosages were 0, 0.5, 5.4, 136, and 275 mg/kg/day
in males, and 0, 0.7, 6.8, 168, and 331 mg/kg/day in females,
respectively over weeks 1-104. Chronic NOEL = 10 ppm (0.5 and
0.7 mg/kg/day in M and F), based on increased
hyaline droplets in kidneys of both sexes at 100 ppm and above,
increased and tubular cell karyomegaly and transitional cell hyperplasia
in kidneys of males, hepatocellular
hypertrophy and increased hepatocellular pigmentation in females,
and calculus of renal pelvis in females. Substantial toxicity
prompted investigators to determine that the two highest dose
levels Òexceeded the maximum tolerated dose and therefore should
not be used for risk assessmentÓ. The study design, with no dose
levels between 100 and 2500 ppm, gave no opportunity to characterize
dose-response in this range of primary interest. These were major
changes at the highest two dose levels (in both sexes, unless
indicated), in addition to the findings for the NOEL (above) applying
to both sexes. Body weights were reduced over time (in males,
primarily in last few weeks of the study), so that at termination
the deficits of 2500 and 5000 ppm groups compared to controls
were 8 and 17% in males, and 18 and 28% in females, respectively.
In females only, this was accompanied by approximately 10% food
consumption reductions at each of the higher dose levels. There
were reductions in the main hematology parameters (RBC counts,
HCT, and Hb levels). Incidence and/or degree of chronic progressive
nephropathy was increased. The majority of these rats had "slight"
to "minimal" degeneration of sciatic nerve and skeletal
muscle (thigh).
Males had elevated incidence of single cell
necrosis in liver. Chronic inflammation
of the lungs was seen in the majority of these rats,
whereas low incidences of congestion of the abdominal cavity,
and urinary bladder hyperplasia were limited to these dose levels.
Numerous clinical chemistry changes mirrored the above pathology
in kidneys,
liver, and perhaps other tissues at the higher two dose
levels. Tumor incidence: thyroid follicular tumors (adenomas and
carcinomas) increased in males (incidences of 1, 1, 1, 7, and
8 in controls through high dose, respectively) and females (incidences
of 0, 0, 1, 5, and 4). Hepatocellular tumors
(primarily adenomas) were increased in males (incidences
of 2, 2, 1, 5, and 11 in controls through high dose, respectively).
There were no statistically significant increases in epithelial
cell tumors in kidneys nor urinary bladder, however the low incidences
of transitional cell papilloma and tubule cell adenoma or carcinoma
were predominantly found in 2500 and 5000 ppm groups (compare
to the congener, trifluralin). Study is acceptable. Tumors are
possible adverse effects, which should be evaluated in perspective
of the many indications of excessive exposures at effective dose
levels. Green and Aldous, 4/11/00.
Ref:
Summary of Toxicological Data: Benefin. California EPA, Department
of Pesticide Regulation, Medical Toxicology Branch. Revised April
21, 2000.
http://www.fluorideaction.org/pesticides/benefin.ca.tox.rev.apr.2000.pdf
Spleen
(click
on for all fluorinated pesticides)
"Benefin: 13-Week Oral
Toxicity Study in Beagle Dogs", (D.W. Dalgard, Hazleton Washington,
HWA 174-135, 12/28/93). Benefin, purity 95.8%, administered via
capsule at concentrations of 0, 5, 25, 125 mg/kg/day to 4 Beagle
dogs/sex/group for 13 weeks. Emesis (compound colored) was slightly
increased at all dose levels for females and for mid and high
dose males. Liver weights was increased for high dose males; and
the incidence of hepatocellular hypertrophy was increased for
high dose males and females. Increased incidence
of hemosiderin pigment was observed in the liver at the
high dose and the spleen at mid and
high dose levels. NOAEL = 5 mg/kg/day. Acceptable (Kishiyama,
J., and P. Iyer, 5/12/98).
Ref: Summary of Toxicology Data for Benefin.
California EPA, Department of Pesticide Regulation, Medical Toxicology
Branch
http://www.cdpr.ca.gov/docs/toxsums/pdfs/53.pdf
Environmental
(click
on for all fluorinated pesticides)
Fish
-- Goldfish (Carassius auratus) - Highly
Toxic
-- Sheepshead
minnow (Cyprinodon variegatus) - Highly
toxic
-- Bluegill
(Lepomis macrochirus) -- Highly
toxic to Very Highly
Toxic
-- Rainbow trout,donaldson
trout (Oncorhynchus mykiss) - Highly
Toxic
-- Fathead
minnow (Pimephales promelas) -Highly
Toxic
Zooplankton
-- Opossum
shrimp (Americamysis bahia) - Very
Highly Toxic
Ref:
PAN Pesticides Database - Chemical Toxicity Studies on Aquatic
Organisms. Toxicity Studies for Benfluralin on All Organism
Groups - Toxicology studies on aquatic organisms from science
journals.
http://www.pesticideinfo.org/List_AquireAll.jsp?Rec_Id=PC35043
Fluoride/fluorinated
substances identified in Agreement between Canada and the
United States on Great Lakes
Water Quality, 1978.
Appendix 1 - Hazardous Polluting Substances:
Ammonium Bifluoride * Ammonium Fluoborate * Ammonium Fluoride
* Ammonium Silicofluoride * Antimony Trifluoride * Beryllium
Fluoride * Ferric Fluoride * Hydrofluoric Acid * Lead Fluoborate
* Lead Fluoride * Sodium Bifluoride * Sodium Fluoride * Zinc
Fluoride * Zinc Silicofluoride * Zirconium Potassium Fluoride.
Appendix 2 - Potential Hazardous Polluting
Substances:
Aluminum Fluoride * Antimony Pentafluoride
* Benfluralin * Chlorflurazole * Cobaltous Fluoride
* Stannous Fluoride |
A
February
15, 2005,
check at the
Code of Federal Regulations for Benfluralin: this herbicide
is permitted in or on 8
food
commodities in the United States.
The
following list identifies these crops for which EPA has
set pesticide tolerances.
|
| [Code
of Federal Regulations]
[Title 40, Volume 22]
[Revised as of July 1, 2004]
From the U.S. Government Printing Office via GPO Access
[CITE: 40CFR180.208]
[Page 378]
TITLE 40--PROTECTION OF ENVIRONMENT
CHAPTER I--ENVIRONMENTAL PROTECTION AGENCY (CONTINUED)
PART 180_TOLERANCES AND EXEMPTIONS FROM TOLERANCES FOR PESTICIDE
CHEMICALS
IN FOOD--Table of Contents
Subpart C_Specific Tolerances
Sec. 180.208 N-Butyl-N-ethyl-[alpha][middot][alpha][middot][alpha]-tri
fluoro-2,6-dinitro-p-toluidine; tolerances for residues
(a) General. Tolerances are established for residues of the
herbicide N-butyl-N-ethyl-[alpha][middot][alpha][middot][alpha]-
trifluoro-2,6-dinitro-p-toluidine in or on the following food
commodities: |
| Commodity |
As
of
December 1,
2003
PPM |
As
of
February 15,
2005
PPM |
CFR |
Alfalfa,
forage
|
Not
Listed in this way |
0.05(N) |
180.208 |
Alfalfa,
hay
|
Not
Listed in this way |
0.05(N) |
180.208 |
| Clover,
forage |
Not
Listed in this way |
0.05(N) |
180.208 |
Clover,
hay
|
Not
Listed in this way |
0.05(N) |
180.208 |
| Lettuce |
0.05 |
0.05(N) |
180.208 |
| Peanut |
0.05 |
0.05(N) |
180.208 |
| Trefoil,
birdsfoot, forage |
Not
Listed in this way |
0.05(N) |
180.208 |
| Trefoil,
birdsfoot, hay |
Not
Listed in this way |
0.05(N) |
180.208 |
ALFALFA
|
0.05 |
Not
Listed in this way |
180.208 |
CLOVER
|
0.05 |
Not
Listed in this way |
180.208 |
TREFOIL,
BIRDSFOOT
|
0.05 |
Not
Listed in this way |
180.208 |
| (b)
Section 18 emergency exemptions. [Reserved]
(c) Tolerances with regional registrations. [Reserved]
(d) Indirect or inadvertent residues. [Reserved] |
|

